-
General Characteristics
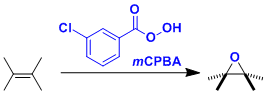
The oxidation of olefins to epoxides using peracids is known as the Prilezhaev epoxidation. m-Chloroperoxybenzoic acid (mCPBA) in particular is used often in laboratories because it is a relatively safe (in terms of explosiveness) solid reagent.
In general, electron-rich olefins are more reactive.
When there is a free stereogenic hydroxyl group near the olefin, the hydrogen bond between the OH group and the reagent leads to diastereoselective epoxidation (the Henbest rule). When the OH group is protected, opposite diastereoselectivity is observed.
-
General References
・Prilezhaev, N. Ber. 1909, 42, 4811. doi:10.1002/cber.190904204100
・Swern, D. Org. React. 1953, 7, 378.
・Henbest Rule: Hoveyda, A. H.; Evans, D. A.; Fu, G. C. Chem. Rev. 1993, 93, 1307. DOI: 10.1021/cr00020a002
-
Reaction Mechanism
The concerted mechanism is generally accepted and the stereochemistry of the alkene is preserved. Electron donating substituents on the alkene promote the reaction.
-
Examples
-
Experimental Procedure
-
Experimental Tips
-
References
-
Related Books

![mCPBA_4[1]](https://assets.en.chem-station.com/uploads/2014/04/mCPBA_41.gif)
![prilezhaev_3[1]](https://assets.en.chem-station.com/uploads/2014/04/prilezhaev_31.gif)
![en-eth1-2[1]](https://assets.en.chem-station.com/uploads/2014/04/en-eth1-21.gif)