- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
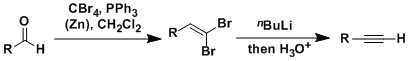
The Corey-Fuchs reaction is the conversion of aldehydes into alkynes with one carbon homologation.
The treatment of 1,1-dihaloalkenes with 2 equivalents of alkyllithium gives lithium acetylides, which give terminal alkynes upon protonation. Doubly substituted alkynes can be synthesized in one-pot by trapping the acetylides with various electrophiles instead of protic acids.
This reaction is obviously not applicable to base sensitive substrates.
-
General References
・Corey, E. J.; Fuchs, P. L. Tetrahedron Lett. 1972, 3769. DOI: 10.1016/S0040-4039(01)94157-7
・Review: Eymery, F.; Iorga, B.; Savignac, P. Synthesis 2000, 185. DOI: 10.1055/s-2000-62
-
Reaction Mechanism
The conversion of aldehydes into 1,1-dibromoalkenes proceeds by the similar mechanism to the Wittig and Appel reactions.
The formation of the alkyne by alkyllithium is not simple E2 elimination. This step is rather understood as the rearrangement of the carbene formed by alpha-elimination of the bromide, which has been confirmed by 13C labelling experiments (Ref: Fritsch-Buttenberg-Wiechell rearrangement).
-
Examples
The synthesis of (-)-laulimalide.[1]
-
Experimental Procedure
-
Experimental Tips
-
References
[1] Ghosh, A. K.; Wang, Y. J. Am. Chem. Soc. 2000, 122, 11027. DOI: 10.1021/ja0027416
-
Related Books

![al-yne1[1]](https://assets.en.chem-station.com/uploads/2014/05/al-yne11.gif)
![corey_fuchs_3[1]](https://assets.en.chem-station.com/uploads/2014/05/corey_fuchs_31.gif)