- Generality
- Reagent Availability
- Experimental User Friendliness
- Application to Other Reactions
- Criteria #5
-
General Characteristics
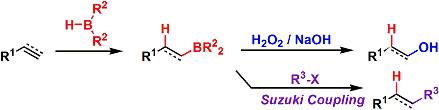
-Alkenes can be converted into anti-Markovnikov alcohols by the Brown hydroboration, which involves regio/stereoselective syn-addition of a B-H bond to the olefin followed by H2O2/NaOH oxidation. The reaction provides a complementary method to oxymercuration and hydration, which give only Markovnikov alcohols. Hydroboration of alkynes give carbonyl compounds.
-The simplest reagent is borane (BH3), which exists as dimeric diborane. But since it is a toxic gas and not particularly reactive either, it is unpractical to be used regularly. In practice, chemists generally use borane complexes (BH3-L) in which the ligand, typically THF, dimethylsulfide, or amine, keeps the borane monomeric. These borane complexes are sold as liquids that can be used easily.
-The problem of controlling regio-, stereo-, and chemoselectivities can be solved by introducing large substituents on the borane. Boron and hydrogen have similar electronegativities, therefore steric hindrance becomes a dominant factor compared with electrostatics. Shown below is a list of common borane reagents. Oxygen-bound catechol borane and pinacol borane are slower to hydroborate and sometimes require assistance from transition metal catalysts.
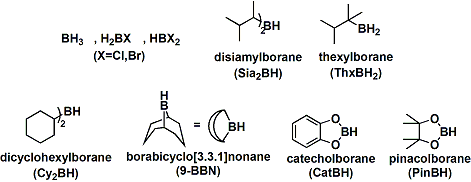
-The synthesis of chiral alcohols is possible using optically active boranes containing chiral substituents. Natural product derived diisopinocampheylborane (Ipc2BH) is particularly useful and finds many applications in large scale syntheses.
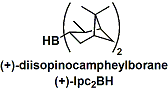
-With the recent progress of palladium catalysis, sequential transformations such as the one linking hydroboration to the Suzuki-Miyaura cross-coupling to make carbon-carbon bonds are now available.
-Herbert Brown won the Nobel Prize in Chemistry in 1979 for his contribution to the advancement of organoboron chemistry, jointly with Geog Wittig.
-
General References
・Brown, H. C.; Subba Rao, B C.J. Am. Chem. Soc. 1956, 78, 5694. DOI: 10.1021/ja01602a063
・Brown, H. C.; Subba Rao, B. C. J. Am. Chem. Soc. 1958, 80, 1552. DOI: 10.1021/ja01540a011
・Brown, H. C.“Hydroboration” W. A. Benjaminem, 1962.
・Zweifel, G. et al. Org. React. 1963, 13, 1.
・Brown, H. C. Tetrahedron 1981, 37, 3547. doi:10.1016/S0040-4020(01)98886-6
・Burgess, K.; Ohlmeyer, M. J. Chem. Rev. 1991, 91, 1179. DOI: 10.1021/cr00006a003
・Beletskaya, I.; Pelter, A. Terahedron 1997, 53, 4957. doi:10.1016/S0040-4020(97)00001-X
-
Reaction Mechanism
1. Addition of B-H bond: An olefin and the empty orbital of boron interact to form a π-complex first. Concerted syn-addition then takes place.
2. Oxidative cleavage of B-C bond: The conjugate base of hydrogen peroxide coordinates with the empty orbital of the boron. The complex then undergoes rearrangement with the elimination of a hydroxyl group. The stereochemistry is retained at the migrating carbon.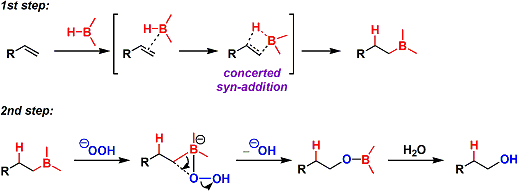
-
Examples
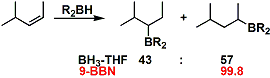
An example of 9-BBN improving regioselectivity.
By replacing H2O2 with either hydroxylamine-O-sulfonate or chloramine, amines can be synthesized.
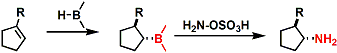
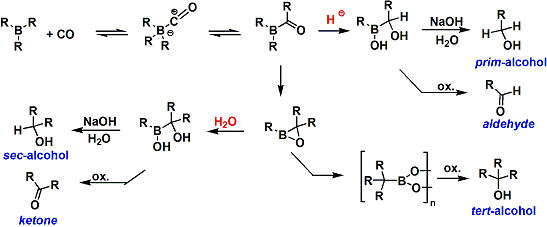
When trialkylborane is heated in the presence of carbon monoxide, all three alkyl groups migrate onto the carbonyl group. Then by hydrolysis, tertiary alcohols can be synthesized. This is an effective way to access extra-bulky alcohols difficult to obtain otherwise. The “bora-epoxide” intermediate is very slow to undergo the third alkyl migration and it can be prevented by adding water to the system. Either secondary alcohols or ketones can be synthesized this way. Additionally, the addition of a hydride source such as LiBH4 leads to the reduction of the “bora-ketone” intermediate after the first alkyl migration, from which either primary alcohols or aldehydes can be synthesized. The summary is shown below.
Hydroboration of terminal alkynes in the presence of a Rh(I) catalyst gives unusual Z-vinylboron products.[1] The reaction is considered to proceed through a rhodium vinylidene intermediate.
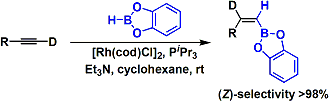
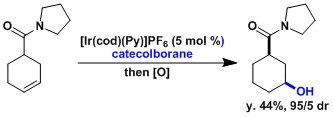
Control/reversal of regioselectivity is possible by choosing the appropriate conditions. In the cases of rhodium catalysis, olefins react faster than ketones. For substrates possessing coordinating groups, hydroboration catalyzed by the Crabtree’s catalyst can be diastereoselective due to the directing effect.
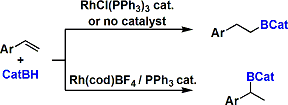
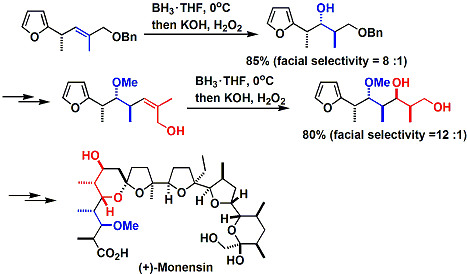
Clever use of stereoselective hydroboration is seen in Kishi’s total synthesis of monensin.[3] The facial selectivity is explained by the addition of the borane from the less hindered side of the alkene in the most stable conformation that avoids the allylic strain.
Hydroboration proceeds at room temperature using pyridine-borane complex. Unlike borane-THF complex, the reaction stops after the first addition. The resulting borane intermediate can be converted into various useful compounds.
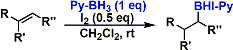
-
Experimental Procedure
-
Experimental Tips
-
References
[1] Ohmura, T.; Yamamoto, Y.; Miyaura, N. J. Am. Chem. Soc. 2000, 122, 4990.DOI:10.1021/ja0002823
[2] (a) Evans, D. A.; Fu, G. C.J. Am. Chem. Soc. 1991, 113, 4042. DOI: 10.1021/ja00010a083 (b) Evans, D. A.; Fu, G. C.; Hoveyda, A. H. J. Am. Chem. Soc. 1992, 114, 6671. DOI: 10.1021/ja00043a009
[3] Schmid, G.; Fukuyama, T.; Akasaka, K.; Kishi, Y. J. Am. Chem. Soc. 1979, 101, 259. DOI: 10.1021/ja00495a064
[4] Clay, J. M.; Vedejs, E. J. Am. Chem. Soc. 2005, 127, 5766. DOI: 10.1021/ja043743j
-
Related Books
[amazonjs asin=”0198556624″ locale=”US” title=”Organic Synthesis: The Roles of Boron and Silicon (Oxford Chemistry Primers)”]