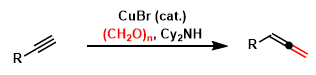- Popularity
- Criteria #2
- Criteria #3
- Criteria #4
- Criteria #5
-
Characteristics
Crabbe allene synthesis (Crabbe homologation) is Cu-cataylzed synthesis of terminal allene compounds via homologation of acetylene precursors in the presence of diisopropylamine and formaldehyde.
-
Literature reference
・Crabbé, P.; Fillion, H.; André, D.; Luche, J.-L. J. Chem. Soc. Chem. Commun. 1979, 859. DOI:10.1039/C39790000859
・Kuang, J.; Ma, S. J. Org. Chem. 2009, 74, 1763. DOI: 10.1021/jo802391x
・Kitagaki, S.; Komizu, M.; Mukai, C. Synlett 2011, 1129. DOI: 10.1055/s-0030-1259936
-
Reaction mechanism
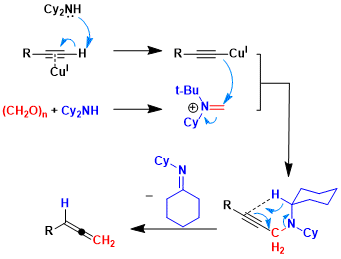
Fillion and coworkers described in their mechanistic study[1] that the Mannich base is observed to form quickly (<15 min) and can be isolated to later be subjected to the retro-ene-like reaction, or it can be left in the reaction flask to afford the allene directly in one pot. Therefore, this step is proven experimentally to be not rate determining. The large experimental KIE observed by Fillion and coworkers also agree with this assumption.
-
Example of reactions
Crabbe and the modification conditions is limited to paraformaldehyde: no allene was formed when other aldehydes were used. In 2010, Ma and coworkers reported new protocol, which uses cost-effective ZnI2 and morpholine affording 1,3-disubstituted allenes from aromatic or aliphatic aldehydes and terminal alkynes.[2a]
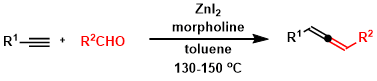
-
Bibliography
[1] Fillion, H. Andre, D. Luche, J.-L. Tetrahedron Lett. 1980, 21, 929. DOI: 10.1016/S0040-4039(00)77741-0
[2] (a) Kuang, J.; Ma, S. J. Am. Chem. Soc. 2010, 132, 1786. DOI: 10.1021/ja910503k (b) Ye, J.; Li, S.; Chen, B.; Fan, W.; Kuang, J.; Liu, J.; Liu, Y.; Miao, B.; Wan, B.; Wang, Y.; Xie, X.; Yu, Q.; Yuan, W.; Ma, S. Org. Lett. 2012, 14, 1346. DOI: 10.1021/ol300296k
-
Related Books
[amazonjs asin=”0471872849″ locale=”US” title=”Allenes in Organic Synthesis”][amazonjs asin=”4431540423″ locale=”US” title=”Total Synthesis of Bioactive Natural Products by Palladium-Catalyzed Domino Cyclization of Allenes and Related Compounds (Springer Theses)”]
-
Related Links
・Come On, Allene ! The Crabbe Reaction (Just like cooking) ・Synthesis of Allene (organic-chemistry.org) ・Synthesis of Substituted Allenes (organic-chemistry.org) ・Allene - Wikipedia