Zárate, C.; Martin, R. J. Am. Chem. Soc. 2014, ASAP.
DOI: 10.1021/ja412107b
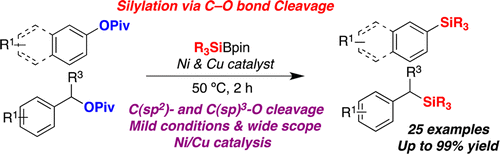
A Ni/Cu-catalyzed silylation of unactivated C–O electrophiles derived from phenols or benzyl alcohols is described. This transformation is characterized by its wide scope and mild conditions, providing a direct access to synthetically versatile silylated compounds. The protocol allows for the coupling of C(sp2)−O and even C(sp3)–O bonds with similar efficiency.
In recent years, the utilization of C−O electrophiles has received considerable attention due to their lack of toxicity and the readily availability of phenol as compared to commonly employed organic halides.[1]
Martin and coworkers reported the discovery of a Ni/Cu catalytic couple that allows for a C− Si bond-forming reaction via cleavage of unactivated C− O bonds in ester derivatives.Various naphthyl and phenol pivalates can be converted to the corresponding silylarenes in the presence of Ni(COD)2 (10 mol %), PCy3 (20 mol %) as a catalyst, CuF2 (30 mol %) and CsF (1 equiv) as additives in toluene at 50 °C. They are suggesting that both Ni and Cu assist the C− O cleavage/C− Si bond-forming event. General procedure is shown below.
General Procedure A (triethyl(naphthalen-2-yl)silane is used as anexample).
An oven-dried 5 mL screw-capped test tube containing a stirring bar was charged with 1a (114 mg, 0.50 mmol) and CuF2 (15.2 mg, 30 mol %). The test tube was introduced in an argon-filled glovebox where Ni(COD)2 (13.8 mg, 10.0 mol %), PCy3 (28.0 mg, 20.0 mol %), CsF (76.0 mg, 1.0 equiv.), a solution of 2a (0.1454 g in 1.50 mL toluene, 1.2 equiv.) and additional toluene (1.5 mL) were then added sequentially. The tube with the mixture was taken out of the glovebox and stirred at 50ºC for 2-15 h. The mixture was then allowed to warm to room temperature, diluted with EtOAc (5 mL) and filtered through a Celite® plug, eluting with additional EtOAc (10 mL). The filtrate was concentrated and purified by column chromatography on silica gel (Hexanes) to yield the title product 3a as a colorless oil in 90% yield (109 mg). Rf 0.65 (Hexanes).
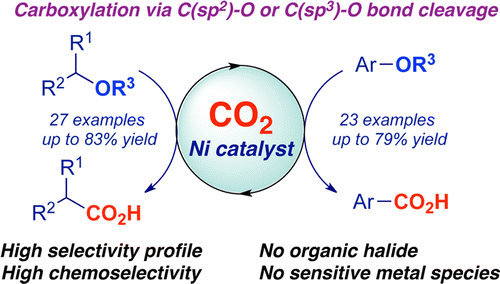
Very recently, Martin has also published the Ni-catalyzed C–O bond transformation to convert aryl carboxylic acid using CO2 as a nucleophiles.[2]
-
References
[1] (a) “Nickel-Catalyzed Cross-Couplings Involving Carbon−Oxygen Bonds”
Rosen, B. M.; Quasdorf, K. W.; Wilson, D. A.; Zhang, N.; Resmerita, A.-M.; Garg, N. K.; Percec, V. Chem. Rev. 2011, 111, 1346. DOI:10.1021/cr100259t
(b) “Recent Progress in Nickel-Catalyzed Biaryl Coupling”
Yamaguchi, J.; Muto, K.; Itami, K. Eur. J. Org. Chem 2013, 19. DOI: 10.1002/ejoc.201200914
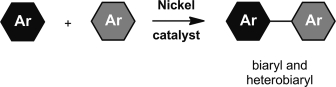
Nickel catalysis for biaryl coupling reactions has received significant attention as a less expensive and less toxic alternative to “standard” palladium catalysis. Here we describe recent developments in nickel-catalyzed biaryl coupling methodology, along with mechanistic studies and applications. In particular we focus on nickel-catalyzed coupling reactions in which “unreactive” bonds such as C–H, C–O, and C–C bonds are converted into biaryl moieties.
[2] “Ni-Catalyzed Carboxylation of C(sp2)– and C(sp3)–O Bonds with CO2”
Correa, A.; León, T.; Martin, R. J Am Chem Soc 2014, 136, 1062. DOI:10.1021/ja410883p
In recent years a significant progress has been made for the carboxylation of aryl and benzyl halides with CO2, becoming convenient alternatives to the use of stoichiometric amounts of well-defined metal species. Still, however, most of these processes require the use of pyrophoric and air-sensitive reagents and the current methods are mostly restricted to organic halides. Therefore, the discovery of a mild, operationally simple alternate carboxylation that occurs with a wide substrate scope employing readily available coupling partners will be highly desirable. Herein, we report a new protocol that deals with the development of a synergistic activation of CO2 and a rather challenging activation of inert C(sp2)–O and C(sp3)–O bonds derived from simple and cheap alcohols, a previously unrecognized opportunity in this field. This unprecedented carboxylation event is characterized by its simplicity, mild reaction conditions, remarkable selectivity pattern and an excellent chemoselectivity profile using air-, moisture-insensitive and easy-to-handle nickel precatalysts. Our results render our method a powerful alternative, practicality and novelty aside, to commonly used organic halides as counterparts in carboxylation protocols. Furthermore, this study shows, for the first time, that traceless directing groups allow for the reductive coupling of substrates without extended π-systems, a typical requisite in many C–O bond-cleavage reactions. Taking into consideration the limited knowledge in catalytic carboxylative reductive events, and the prospective impact of providing a new tool for accessing valuable carboxylic acids, we believe this work opens up new vistas and allows new tactics in reductive coupling events.
-
Related Links
Ruben Martin Group (ICIQ, Spain)
-
Related Books
[amazonjs asin=”3527332790″ locale=”US” title=”Transition Metal-Catalyzed Couplings in Process Chemistry: Case Studies From the Pharmaceutical Industry”][amazonjs asin=”3527331549″ locale=”US” title=”Metal Catalyzed Cross-Coupling Reactions and More, 3 Volume Set”]