- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
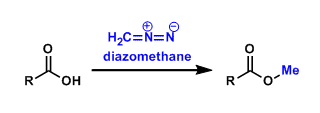
Diazomethane (CH2N2) is the simplest example of diazo compounds.
It is traditionally used to synthesize methyl esters of carboxylic acids. This methyl ester formation is very fast (usually done within 5 minutes) and alcohols are not methylated by this reagent.
Diazomethane is also used for the Ardnt-Eistert synthesis (the Wolff rearrangement).
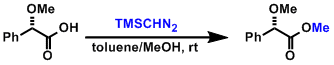
It is important to recognize that diazomethane is a potentially dangerous reagent due to its explosive and carcinogenic properties. Trimethylsilyldiazomethane, which is sold as a solution in hexanes, is less explosive and often used as a substitute.
-
General References
- Moore, J. A.; Reed, D. E. Org. Synth. 1961, 41, 16. [website]
- Shioiri, T.; Aoyama, T.; Mori, S. Org. Synth., Coll. Vol. 1993, 8, 612.
- Aoyama, T.; Shioiri, T. Tetrahedron Lett. 1980, 21, 46. DOI:10.1016/S0040-4039(00)92200-7
-
Reaction Mechanism
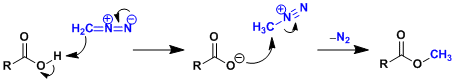
-
Examples
-
Experimental Procedure
In a standard procedure, diazomethane is prepared from Diazaid®.

To minimize explosion risks, use the glassware with clear-seal joints available from Aldrich for the preparation of diazomethane.

(Picture: from Wikipedia)
-
Experimental Tips
・Preparation of diazomethane in high concentrations can lead to explosion accidents. When one has to use it, use it on small scales and as dilute solutions in ether.
・Since the solutions of diazomethane are colored yellow, one can tell the consumption of diazomethane by the disappearance of the color.
・Diazomethane should not touch sharp edges, metal powders, normal ground-glass joints, and flasks with scratches. These can trigger detonation. For transferring diazomethane solutions, use pipettes with flame-dulled tips.
・Diazomethane and its precursors are strongly carcinogenic. Handle them under well-ventilated hoods.
-
References
-
Related Reactions
- Regitz Diazo Transfer
- Protection of Carboxylic Acid
- Cyclopropanation with Metal Carbenoid
- C-H Insertion of Metal Carbenoid
- Seyferth-Gilbert Alkyne Synthesis
- Arndt-Eistert Synthesis
- Wolff Rearrangement
-
Related Books
-
External Links
- diazomethane – Wikipedia
- Trimethylsilyldiazomethane– Wikipedia
- Diazald (Sigma-Aldrich)