- Generality
- Reagent Availability
- Experimental User Friendliness
- Scope of Reported Examples
- Criteria #5
-
General Characteristics
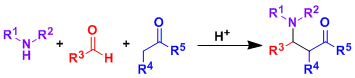
-The Mannich reaction is a classic example of three-component condensation reactions.
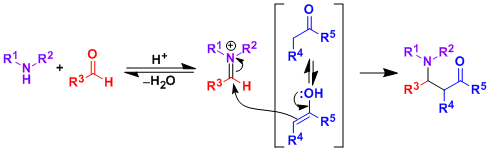
-Under acidic conditions, an amine and an aldehyde condense to form an iminium ion, which is attacked by an enolizable ketone. The Mannich reaction is an important carbon-carbon bond forming synthetic method.
-It is common to use preformed imines and react it with a ketone nucleophile (especially for stereoselective cases).
-
General References
・Mannich, C.; Krosche, W. Arch. Pharm. 1912, 250, 647.
・Brewster, J. H. et al. Org. React. 1953, 7, 99.
・Blicke, F. F. Org. React. 1942, 1, 303.
・Merz, K. W. Pharmazie 1956, 11, 505.
・Tramontini, M.; Tetrahedron 1990, 46, 1791. doi:10.1016/S0040-4020(01)89752-0
・Kleinman, E. F. Comp. Org. Syn. 1991, 2, 893.
・Heane, H. Comp. Org. Syn. 1991, 2, 953.
・Overman, L. E.; Ricca, D. J. Comp. Org. Syn. 1991, 2, 1007.
・Arend, M.; Westermann, B.; Risch, N. Angew. Chem. Int. Ed. 1998, 37, 1044. [abstract]
・Kobayashi, S.; Ishitani, H. Chem. Rev. 1999, 99, 1069. DOI: 10.1021/cr980414z
-
Reaction Mechanism
-
Examples
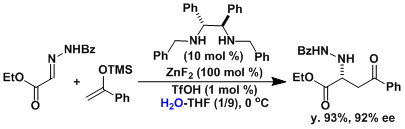
The Mannich reaction is useful in synthesizing β-aminocarbonyl compounds, which are substructures commonly found in biologically active compounds. A number of asymmetric Mannich reactions have been developed. For example, Kobayashi developed the catalytic systems that can work in water.[1]
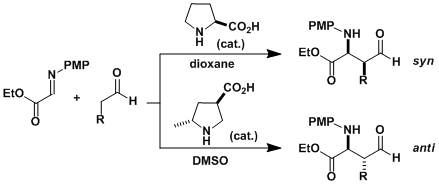
L-Proline catalyzes the enantioselective addition of aldehydes to imines.[2a] The proline analogue containing the carboxylic acid at a different position also catalyzes the reaction but with different relative stereochemistry.[2b] See the List-Barbas aldol reaction as reference.
-
Experimental Procedure
-
Experimental Tips
-
References
[1] Kobayashi, S.; Hamada, T.; Manabe, K. J. Am. Chem. Soc. 2002, 124, 5640. DOI: 10.1021/ja026094p
[2] (a) Barbas, C F., III et al. J. Am. Chem. Soc. 2002, 124, 1866. DOI:10.1021/ja017833p (b) Barbas, C. F., III et al. J. Am. Chem. Soc. 2006, 128, 1040. DOI: 10.1021/ja056984f
-
Related Books
[amazonjs asin=”3527308067″ locale=”US” title=”Multicomponent Reactions”]
[amazonjs asin=”3527305173″ locale=”US” title=”Asymmetric Organocatalysis: From Biomimetic Concepts to Applications in Asymmetric Synthesis”]
[amazonjs asin=”3540734945″ locale=”US” title=”Organocatalysis (Ernst Schering Foundation Symposium Proceedings)”]
[amazonjs asin=”0471298050″ locale=”US” title=”Catalytic Asymmetric Synthesis, Second Edition”]