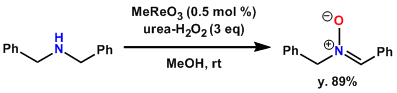Overall Score4
- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
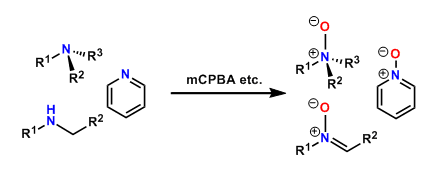
Amines are susceptible to oxidation. Tertiary amines are oxidized to give N-oxides.
Secondary amines having proton(s) at the α-position are oxidized to nitrones. The conversion to hydroxylamines is possible depending on the conditions.
The oxidation of heterocyclic amines such as pyridine requires more forcing conditions. In these cases, oxidants such as trifluoroperacetic acid and methyltrioxorhenium(VII) are effective.
-
General References
-
Reaction Mechanism
-
Examples
-
Experimental Procedure
Synthesis of a nitrone using catalytic MeReO3.[1]
A 250-mL, two-necked, round-bottomed flask equipped with a magnetic stirbar, thermometer, and a reflux condenser fitted with a rubber septum and balloon of argon is charged with a solution of methyltrioxorhenium (MTO) (0.013 g, 0.05 mmol, 0.1% mol equiv) in 100 mL of methanol. Urea hydrogen peroxide (UHP) (14.3 g, 152 mmol) is added, the flask is cooled in an ice bath, and dibenzylamine (9.7 mL, 50.7 mmol) is then added dropwise via syringe over 10 min. After completion of the addition, the ice bath is removed and the mixture is stirred at room temperature. A white precipitate forms after approximately 5 min and the yellow color disappears within 20 min. Another four portions of MTO (0.1% mol equiv, 0.013 g each) are added at 30-min intervals (2.5 hr total reaction time). After each addition, the reaction mixture develops a yellow color, which then disappears; only after the last addition does the mixture remain pale yellow. The reaction flask is cooled in an ice bath and solid sodium thiosulfate pentahydrate (12.6 g, 50.7 mmol) is added in portions over 20 min in order to destroy excess hydrogen peroxide. The cooled solution is stirred for 1 hr further, at which point a KI paper assay indicates that the excess oxidant has been completely consumed. The solution is decanted into a 500-mL flask to remove small amounts of undissolved thiosulfate. The solid is washed with 50 mL of MeOH and the methanol extract is added to the reaction solution which is then concentrated under reduced pressure by rotary evaporation. Dichloromethane (250 mL) is added to the residue and the urea is removed by filtration through cotton and celite. Concentration of the filtrate affords 10.3 g (97%) of the nitrone as a yellow solid. If desired, the nitrone can be recrystallized from diisopropyl ether (200 mL), affording 9.0 g (84%) of a white solid. Concentration of the mother liquor and recrystallization of the residue from 40 mL of diisopropyl ether provides 0.600 g of additional nitrone. Total yield: 9.6 g (89%) of the nitrone as a white solid.
-
Experimental Tips
-
References
[1] Goti, A.; Cardona, F.; Soldaini, G. Org. Synth. 2005, 81, 204. [PDF]
-
Related Reactions
-
Related Books
-
External Links