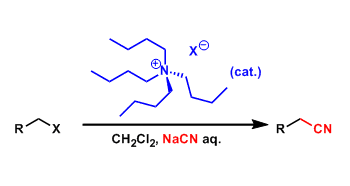
- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
Reactions such as SN2 substitution involve deprotonation by strong bases and combining the resulting anions with electrophiles. These reactions often necessitate the use of polar nonprotic solvents such as DMF and DMSO in order to increase the reactivity and the solubility. These solvents have high boiling points and are relatively expensive, thus are not ideal for large scale applications.
Phase-transfer catalysts (PTC) are used to promote ionic reactions like SN2 substitution in biphasic systems consisting of water and nonpolar organic solvents. These catalysts can move across the two layers and are called amphiphilic. Famous examples are quaternary ammonium salts possessing long alkyl groups and crown ethers.
The advantages of using PTC include use of inexpensive and easy-to-workup inorganic bases, reduction of organic solvents, avoidance of DMF and DMSO, simplification of experiments, improvement of reactivity, and suppression of side reactions, all of which are important especially on large scales. PTC’s are therefore considered a promising technology from green chemistry point of view.
-
General References
- Makosza, M.; Serafinowa, B. Rocz. Chem. 1965, 39, 1223.
- Starks, C. M. J. Am. Chem. Soc. 1971, 93, 195. DOI: 10.1021/ja00730a033
- Dolling, U.-H.; Davis P; Grabowski, E. J. J. Am. Chem. Soc. 1984, 106, 446. DOI: 10.1021/ja00314a045
- Makosza, M. Pure Appl. Chem. 2000, 72, 1399. doi:10.1351/pac200072071399
- O’Donnell, M. J. Acc. Chem. Res. 2004, 37, 506. DOI: 10.1021/ar0300625
- Hashimoto, T.; Maruoka, K. Chem. Rev. 2007, 107, 5656. doi:10.1021/cr068368n
- Ooi, T.; Maruoka, K. Aldrichimica Acta 2007, 40, 77. [PDF]
-
Reaction Mechanism
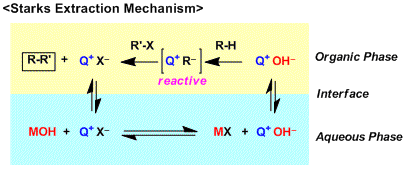
For systems that consist of inorganic bases and quaternary ammonium salts, the mechanism is still a debatable subject but there are two major theories.
The first theory is the Extraction Mechanism (J. Am. Chem. Soc. 1971, 93, 195.) proposed by Starks, which is based on the hypothesis that PTC (denoted as Q+X– below) can move freely across organic and aqueous layers. The inorganic base (MOH) and PTC undergo ion exchange and the resulting Q+OH– is extracted into the organic layer. The Q+OH– species, which is highly polarized and thus is highly basic, deprotonates the reactant. The deprotonated species (Q+R–) is also a highly polarized species and considered highly reactive.
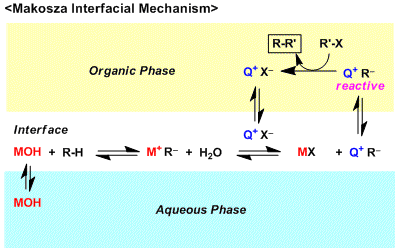
The second theory is the Interfacial Mechanism (Rocz. Chem. 1965, 39, 1223.) proposed by Makosza and it is based on the hypothesis that the deprotonation and the anion generation processess occur at the interface of two layers. According to this theory, PTC does not participate in these processes and plays a role in only the extraction of the reactive species. Asymmetric PTC systems are particularly considered to follow this mechanism, since many of asymmetric PTC’s contain lipophilic groups that make it difficult for them to go into the aqueous layer freely.
-
Examples
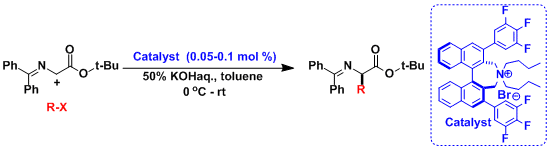
A typical example of rate acceleration by PTC.

The asymmetric phase-transfer catalysis by the Maruoka catalyst: Protected glycine was alkylated enantioselectively using very small amounts of the catalyst based on chiral quaternary ammounium salt.[1]
-
Experimental Procedure
-
Experimental Tips
-
References
[1] Kitamura, M.; Shirakawa, S.; Maruoka, K. Angew. Chem. Int. Ed. 2005, 44, 1549. doi:10.1002/anie.200462257
-
Related Reactions
-
Related Books
[amazonjs asin=”0841234914″ locale=”US” title=”Phase-Transfer Catalysis: Mechanisms and Syntheses”]
[amazonjs asin=”9401042977″ locale=”US” title=”Phase-Transfer Catalysis: Fundamentals, Applications, and Industrial Perspectives”]
[amazonjs asin=”0123884756″ locale=”US” title=”Quaternary Ammonium Salts: Their Use in Phase-Transfer Catalysis”]
-
External Links
- Phase-Transfer Catalyst – Wikipedia
- 京都大学大学院理学系研究科 丸岡研究室 (Kyoto University, Maruoka Laboratory)
- The Industrial Phase-Transfer Catlaysis Website