- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
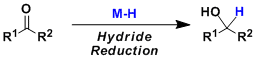
Carbonyl compounds are reduced by metal hydride reagents to give alcohols. A number of reagents with various reducing strengths and properties are known. The metal hydride reagents listed below are used commonly based on practical qualities such as availability and ease of handling.
-
Types
NaBH4 (Sodium Borohydride)
NaBH4 is one of the most widely used reducing agents. It is stable to moisture and air and used in industrial applications. Methanol or ethanol is used as the solvent for the solubility reason. It cannot reduce esters, amides, and carboxylic acids, but esters containing a heteroatom at the α-position are the exceptions that can be reduced (due to the neighboring group assistance). 1,4-Reduction is generally faster for α,β-unsaturated carbonyl compounds, but 1,2-reduction is possible by the addition of cerium salts (the Luche reduction).
LiAlH4 (Lithium Aluminum Hydride or LAH)
LiAlH4 is a strong reducing agent capable of reducing ketones, aldehydes, and even carboxylic acids and esters into alcohols. It can reduce nitriles and amides to amines as well. It also reacts with organic halides and sulfonates to displace them with hydrides. Epoxides are also reduced to give ring-opened products.
THF or diethylehter is used as the inert solvent that dissolves LAH. Water and protic solvents react violently with LAH to generate hydrogen gas. The high reactivity combined with easy-to-scatter fluffiness of the reagent often cause fire accidents. One must be careful when handling this reagent.
(i-Bu)2AlH (Diisobutyl Aluminum Hydride or DIBAL)
DIBAL is Lewis acidic because it has an empty orbital on the aluminum, thus shows different reactivities compared with the “ate” complexes like LAH.
It reduces nitriles to imines, which can be hydrolyzed to aldehydes. Therefore, nitriles can be thought of as the precursory equivalent of aldehydes.
Acetals can be reduced to ethers. For example, treating benzylidene acetals with DIBAL leads to products in which the more congested position is protected as a Bn ether. (See PMB protection for reference.)
The reduction of esters to aldehydes is possible at low temperatures but preventing the overreduction is generally not easy. Stepwise sequence of complete reduction to alcohols and then oxidizing them back to aldehydes is more reliable. As an exception, the partial reduction of five- or six-membered lactones to lactols is easy. It has been reported recently that the ate complex formed by the addition of NaOtBu works as an effective partial reducing agent.
LiAlH(i-Bu)2(n-Bu), another ate complex formed by adding n-BuLi, is a strong reducing agent with unique reactivities.
NaBH3CN (Sodium Cyanoborohydride)
NaBH3CN is a weaker reducing agent than NaBH4 but is stable under acidic conditions (up to pH~3). It is therefore used frequently for the reduction of iminium cations under acidic conditions (Borch reductive amination).
LiBH4 (Lithium Borohydride)
LiBH4 is more strongly reducing than NaBH4 and capable of reducing esters to alcohols. It is sold as solutions but can also be prepared in situ from LiCl and NaBH4.
LiBHEt3 (Lithium Triethylborohydride or Super-Hydride)
LiBHEt3 is an extremely strong reducing agent sold by the tradename Super-Hydride. It is often used for the displacement of halides and sulfonates by hydrides.
BH3•L (Borane Complex)
Borane-dimethylsulfide (BH3•SMe2) and borane-THF (BH3•THF) are famous among commercially available borane complexes. These complexes are easier to use than toxic and gaseous diborane and thus used widely. They are capable of reducing carboxylic acids selectively over ketones. Note that they also hydroborate olefins.
Et3SiH (Triethylsilane)
While the reaction conditions for most reductions are basic, the reduction using Et3SiH is done under acidic conditions (e.g. w/TFA). It finds applications in the reduction of acetals and the reductive removal of alkoxy groups from the anomeric position of sugars. It is also used as a hydrosilylation reagent in combination with transition metal catalysts.
Sodium Bis(2-mehoxyethoxy)Aluminum Hydride (Red-Al)
Red-Al has similar reducing properties to LAH, but it is less prone to ignition and more amenable to large scale applications.
The following reagents are used for more specific purposes.
Ni(BH4)2 (Nickel Borite)
Ni(BH4)2 can reduce nitriles and nitro groups and is prepared in situ from NiCl2 and NaBH4.
NaBH(OAc)3 (Sodium Tri(acetoxy)borohydride)
Other than for reductive amination, this reagent is used to synthesize anti-1,3-diols from β-hydroxy carbonyl compounds. The diastereoselectivity is based on the internal hydride delivery within the six-membered chiarlike transition state.
Zn(BH4)2 (Zinc Borohydride)
The chelating ability of zinc allows for syn-diastereoselective reduction of β-ketoesters. It also reduces β-hydroxy carbonyl compounds to syn-1,3-diols, providing a complementary method to NaBH(OAc)3.
LiBH(s-Bu)3 (Lithium Tri(sec-butyl)borohydride or L-Selectride),
KBH(s-Bu)3 (Potassium Tri(sec-butyl)borohydride or K-Selectride)
These are strong and bulky reducing agents. Their reactivity is highly sensitive to steric hindrance of the substrates.
Cp2ZrHCl or Schwartz Reagent
Cp2ZrHCl is a reagent for hydrozirconation of alkenes and alkynes. The resulting organozirconium compounds can be elaborated further by cross coupling reactions. It is also used as a catalyst for the hydroboration of less reactive alkenes. It can reduce lactams to cyclic imines too.
[(Ph3P)CuH]6 or the Stryker Reagent
The Stryker reagent is used for 1,4-reduction of α,β-carbonyl compounds. Asymmetric reduction is possible using chiral bidentate ligands.
(n-Bu)3SnH (Tributyltin Hydride)
Tributyltin hydride is normally used with a radical initiator for the Barton-McCombie-type reactions. In the presence of transition metal catalysts such as palladium, it can undergo transmetallation and hydrostannation, or form other metal hydride species.
-
General References
-
Reaction Mechanism
-
Examples
Regioselective reduction of malic acid ester.[1]
Diastereoselective reduction of a β-hydroxyketone using Me4NHB(OAc)3[2]: More diastereoselective than NaHB(OAc)3.
One-step conversion of a nitrile into a Boc-protected amine[3a]: The combination of a Lewis acid and NaBH4 can also reduce nitro, azide, and imine groups to amines.
-
Experimental Procedure
-
Experimental Tips
-
References
[1] Saito, S. et al. Tetrahedron 1992, 48, 4067. doi:10.1016/S0040-4020(01)92187-8
[2] Evans, D. A.; Chapman, K. T.; Carreira, E. M. J. Am. Chem. Soc. 1988, 110, 3560. DOI: 10.1021/ja00219a035
[3] (a) Caddick, S.; Judd, D. B.; Lewis, A. K. de K.; Reich, M. T.; Williams, M. R. V. Tetrahedron 2003, 59, 5417. doi:10.1016/S0040-4020(03)00858-5 (b) Chary, K. P.; Ram, S. R.; Iyengar, D. S. Synlett 2000, 683. DOI: 10.1055/s-2000-6614 (c) Cho, B. T.; Kang, S. K. Synlett 2004, 1484. DOI: 10.1055/s-2004-829066 (d) Fringuelli, F.; Pizzo, F.; Vaccaro, L. Synthesis 2000, 646. DOI: 10.1055/s-2000-6389
-
Related Books

![H_red_4[1]](https://en.chem-station.com/wp-content/plugins/lazy-load/images/1x1.trans.gif)