K. Shima, K. Mutoh, Y. Kobayashi, J. Abe, J. Am. Chem. Soc. 2014, 136, 3796–3799.
DOI: 10.1021/ja501028v
The widely tunable optical properties and the visible sensitivity have been required for fast photochromic molecules whose coloration–decoloration cycle completes in μs to ms time scale not only for practical applications such as full-color holographic displays but also for fundamental researches in biochemistry. However, the so far developed [2.2]paracyclophane-bridged imidazole dimers, which are one of the best candidates for fast photochromic molecules, have their weaknesses for these requirements. Herein, we overcome the issues with sustaining fast photochromism and high durability by flipping the two imidazole rings (the head-to-tail and tail-to-tail forms). The alteration in the relative configuration of the imidazole rings suppresses the broad absorption band resulting from the radical–radical interaction. The substitution to the 2-position of the imidazole ring of the tail-to-tail form gives the drastic changes in the steady-state and the transient absorption spectra. The pyrene-substituted tail-to-tail form demonstrates that the transient absorption spectrum is featured by the inherent spectrum of the imidazolyl radical. This molecular framework is easy to functionalize fast photochromic molecules such as sensitizations to the red light, chirality, and biological tagging, and therefore it is versatile for various fast photochromic applications.
Photochromic molecules which reversely react upon light irradiation such as diarylethenes, have interested in their applications including optical waveguides and shutters, optical data storage, and ophthalmic plastic lenses.
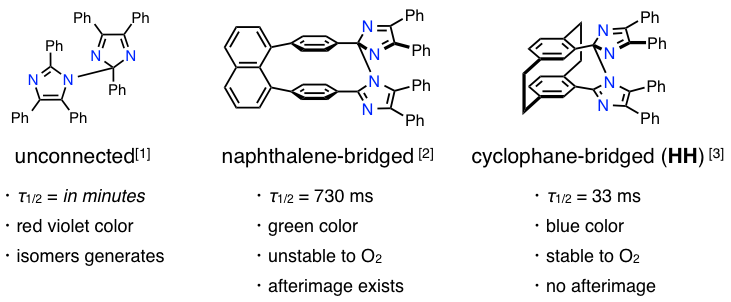
Imidazole dimmers are also known as a kind of photochromic molecules. Hexaarylbiimidazole (HABI), shown in below, was discovered by Hayashi and Maeda,[1] and it has subsequently attracted significant interest because of its unusual physical properties. Various stimuli, such as heat, light, and pressure, readily cleave HABI into a pair of 2,4,5-triphenylimidazolyl radicals (TPIRs), which thermally recombine to reproduce their original imidazole dimer. The solution of HABI changes from colorless to purple under UV irradiation.
In 2008, Prof. Abe, Aoyama Gakuin Universitry, have successfully developed naphthalene-bridged HABI, which is improved photochromic performance in coloration and thermal bleaching rates as well as a greater optical density in the colored state.[2] They cal it fast photochlromic molecules, that can potentially be applied to real-time image processing at video frame rates. However, it was unstable for O2 and needed to be improved. In 2009, Abe and coworkers can be improved the structure by changing cyclophane-bridged structure, which was stable to O2 and the much faster photochromic molecules.[3] You can see the movie in YouTube.
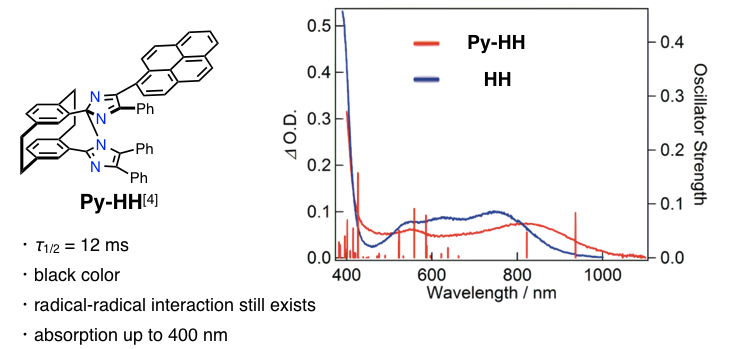
More recently, to enhance the photosensitivity, they introduced pyrenyl moieties to the [2.2]paracyclophane-bridged imidazole dimer as shown in below.[4]
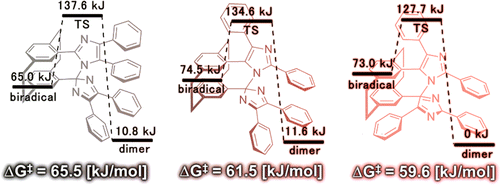
In this paper, they designed the head-to-tail and tail-to-tail forms of the bridged imidazole dimer and systematically analyzed the effect of the relative configuration of the imidazole rings on the photochromic properties. Py-TT achieved visible sensitizations, and no radical− radical interactions were confirmed in the absorption spectrum of the colored species, which offers the tunable transient color selectivity from blue to red.
-
References
[1] Hayashi, T.; Maeda, K. Bull. Chem. Soc. Jpn. 1960, 33, 565. DOI: 10.1246/bcsj.33.565
[2] Fujita, K.; Hatano, S.; Kato, D.; Abe, J. Org. Lett. 2008, 10, 3105. DOI:10.1021/ol801135g
[3] “A Fast Photochromic Molecule That Colors Only under UV Light”
Kishimoto, Y.; Abe, J. J. Am. Chem. Soc. 2009, 131, 4227. DOI: 10.1021/ja810032t
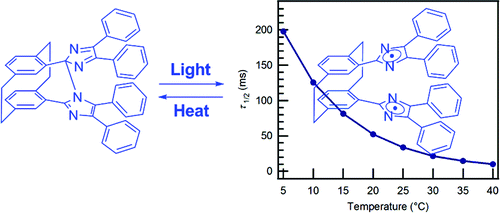
We demonstrate that photochromism based on light-driven reversible bond cleavage can enable rapid coloration upon UV light irradiation and successive fast thermal bleaching within tens of milliseconds at room temperature. We have succeeded in developing a bisimidazole derivative with a paracyclophane moiety, pseudogem-bisDPI[2.2]PC, that changes from colorless to blue when irradiated with UV light. The incident light homolytically cleaves the C−N bond that links the two imidazole rings, generating a pair of imidazolyl radicals. A paracyclophane moiety connects the two radicals, thereby preventing them from diffusing away from one another, and the radicals quickly bind with each other to flip back to the original bisimidazole. Photochromic materials showing such intense photocoloration and fast thermal bleaching performance could be promising materials for possible fast light modulator applications.
[4] Yamashita, H.; Abe, J. J. Phys. Chem. A 2011, 115, 13332. DOI: 10.1021/jp204440s
-
Movie
-
Related goods and books
[amazonjs asin=”B007JZ1TZO” locale=”US” title=”3M Smart Lens Protective Eyewear, 13407-00000-5 Photochromatic Lens, Black Frame (Pack of 1)”] [amazonjs asin=”3642018653″ locale=”US” title=”Molecular Organometallic Materials for Optics (Topics in Organometallic Chemistry)”]
-
Related Links