- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
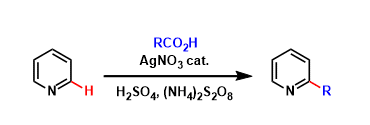
The Minisci reaction allows for the direct functionalization of electron deficient hetero aromatic rings with carbon radicals. The radical precursor is most commonly the combination of carboxylic acid and one electron oxidant, but other C-H bond activation conditions can be used as well.
-
General References
- Minisci, F.; Bernardi, R.; Bertini, F.; Galli, R.; Perchinummo, M. Tetrahedron 1971, 27, 3575. doi:10.1016/S0040-4020(01)97768-3
- Minisci, F.; Vismara, E.; Fontana, F.; Morini, G.; Serravalle, M.; Giordano, C. J. Org. Chem. 1987, 52, 730. DOI: 10.1021/jo00381a004
- Review; Minisci, F.; Vismara, E.; Fontana, F. Heterocycles 1989, 28, 489. DOI: 10.3987/REV-88-SR1
- Review: Duncton, M. A. J. Med. Chem. Commun. 2011, 2, 1135. DOI: 10.1039/C1MD00134E
-
Reaction Mechanism
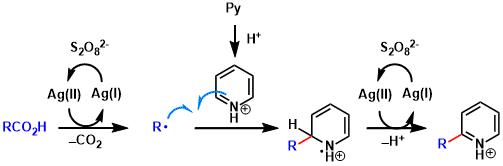
In the generally accepted mechanism, the radical adds to the protonated electrophilic heteroaromatic ring. Acylated aromatic rings tend to form as byproducts. (Ref: J. Org. Chem. 1991, 56, 2866.)
-
Examples
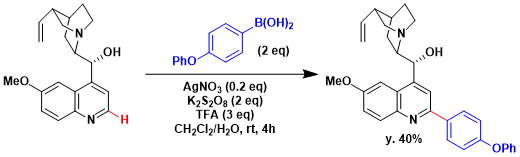
Baran reported a modified procedure in which boronic acids are used instead of carboxylic acids. The room temperature, aqueous, and open-air conditions make this reaction highly practical.[1]
-
Experimental Tips
-
References
[1] (a) Seiple, I. B.; Su, S.; Rodriguez, R. A.; Gianatassio, R.; Fujiwara, Y.; Sobel, A. L.; Baran, P. S. J. Am. Chem. Soc. 2010, 132, 13194. DOI: 10.1021/ja1066459 (b) Fujiwara, Y.; Domingo, V.; Seiple, I. B.; Gianatassio, R.; Bel, M. D.; Baran, P. S. J. Am. Chem. Soc. 2011, 133, 3292. DOI: 10.1021/ja111152z (c) Lockner, J. W.; Dixon, D. D.; Risgaard, R.; Baran, P. S. Org. Lett. 2011, 13, 5628. DOI: 10.1021/ol2023505 (d) Dixon, D. D.; Lockner, J. W.; Zhou, Q.; Baran, P. S. J. Am. Chem. Soc. 2012, 134, 8432. DOI: 10.1021/ja303937y
-
Related Reactions
-
Related Books
[amazonjs asin=”3642123554″ locale=”US” title=”C-H Activation (Topics in Current Chemistry)”]
[amazonjs asin=”3642084362″ locale=”US” title=”Activation of Unreactive Bonds and Organic Synthesis (Topics in Organometallic Chemistry)”]
-
External Links