- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
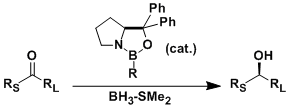
The asymmetric reduction of ketones catalyzed by the chiral oxazaborolidine (derived from proline) is called the Corey-Bakshi-Shibata (CBS) reduction. The reaction has a wide substrate scope including both linear and cyclic ketones, converting them into corresponding alcohols with high enantioselectivity.
The reaction times are short and the experimental procedure is also simple. The reaction usually works simply by mixing the reagents with control of the temperature.
Notably, α,β-unsaturated ketones undergo 1,2-reduction under the CBS conditions.
-
General References
・Corey, E. J.; Bakshi, R. K.; Shibata, S. J. Am. Chem. Soc. 1987, 109, 5551. DOI: 10.1021/ja00252a056
・Corey, E. J.; Shibata, S.; Bakshi, R. K. J. Org. Chem. 1988, 53, 2861. DOI: 10.1021/jo00247a044
・Catalyst Preparation: Mathre et al. J. Org. Chem. 1993, 58, 2880. DOI: 10.1021/jo00062a037
・Singh, V. K. Synthesis 1992, 605. DOI: 10.1055/s-1992-26174
・Deloux, L.; Srebnik, M. Chem. Rev. 1993, 93, 763. DOI: 10.1021/cr00018a007
・Corey, E. J.; Helal, C. J. Angew. Chem. Int. Ed. 1998, 37, 1986. [abstract]
-
Reaction Mechanism
The dual-activation mechanism has been proposed based on crystal structure analysis of the oxazaborolidine-borane complex. As shown below, the Lewis acidic boron atom of the catalyst activates the ketone and the Lewis basic nitrogen atom activates the borane reagent. (Ref: J. Org. Chem. 1993, 58, 799)
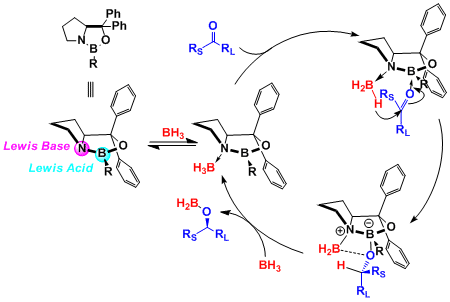
-
Examples
Chiral allylic alcohols can be thought of as precursors of stereogenic carbons via various carbon-carbon bond forming reactions. For example, the sequence of the CBS reduction followed by the Ireland-Claisen rearrangement was used to synthesize a challenging quaternary stereogenic carbon in the synthesis of aspidophytine.[1]
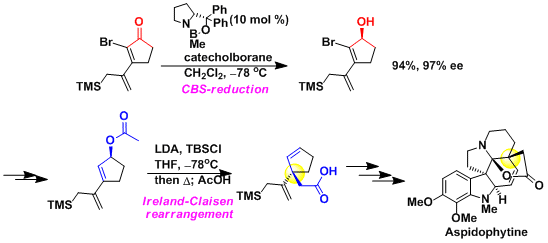
-
Experimental Procedure
-
Experimental Tips
-
References
[1] He, F.; Bo, Y.; Altom, J. D.; Corey, E. J. J. Am. Chem. Soc. 1999, 121, 6771. DOI: 10.1021/ja9915201
-
Related Books
[amazonjs asin=”0471298050″ locale=”US” title=”Catalytic Asymmetric Synthesis, Second Edition”]