- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
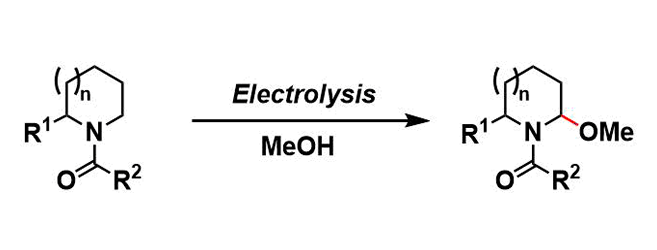
In the Shono oxidation, amides or carbamates in alcohol solvents are electrochemically oxidized to give N,O-acetals. This electrochemical reaction provides a useful way to activate and functionalize the α-position of amines.
-
General References
- Shono, T.; Hamaguichi, H.; Matsumura, Y. J. Am. Chem. Soc. 1975, 97, 4264. DOI: 10.1021/ja00848a020
- Shono, T.; Matsumura, Y.; Tsubata, K. Tetrahedron Lett. 1981, 22, 3249. doi:10.1016/S0040-4039(01)81876-1
- Shono, T.; Matsumura, Y.; Tsubata, K. J. Am. Chem. Soc. 1981, 103, 1172. DOI: 10.1021/ja00395a029
- Shono, T.; Matsumura, Y.; Tsubata, K. Org. Synth. 1984, 63, 206. DOI: 10.15227/orgsyn.063.0206
- Shono, T.; Matsumura, Y.; Uchida, K.; Tsubata, K.; Makino, A. J. Org. Chem. 1984, 49, 300. DOI: 10.1021/jo00176a016
- Shono, T.; Matsumura, Y.; Tsubata, K.; Uchida, K.; Kanazawa, T.; Tasuda, K. J. Org. Chem. 1984, 49, 3711. DOI: 10.1021/jo00194a008
- Alfonso-Súarez, P.; Kolliopoulos, A. V.; Smith, J. P. Banks, C. E.; Jones, A. M. Tetrahedron Lett. 2015, 56, 6863. doi:10.1016/j.tetlet.2015.10.090
Reviews
- Shono, T. Tetrahedron 1984, 40, 811. doi:10.1016/S0040-4020(01)91472-3
- Onomura, O. Heterocycles 2012, 85, 2111. DOI: 10.3987/REV-12-744
- Jones, A. M.; Banks, C. E. Beilstein J. Org. Chem. 2014, 10, 3056. doi: 10.3762/bjoc.10.323
-
History
The reaction was developed by Professor Tatsuya Shono and his colleagues at Kyoto University.
-
Reaction Mechanism
Electrochemical anodic oxidation generates N-acyliminium intermediate, which is trapped by alcohol solvent. When performed in nonalcoholic solvents at low temperature, N-acyliminium ion can be pooled and reacted with various nucleophiles (known as the Cation Pool method).

-
Examples
Among organic electrochemical reactions, the Shono oxidation is one of the reactions that have been well-studied and applied to the synthesis of complex molecules.
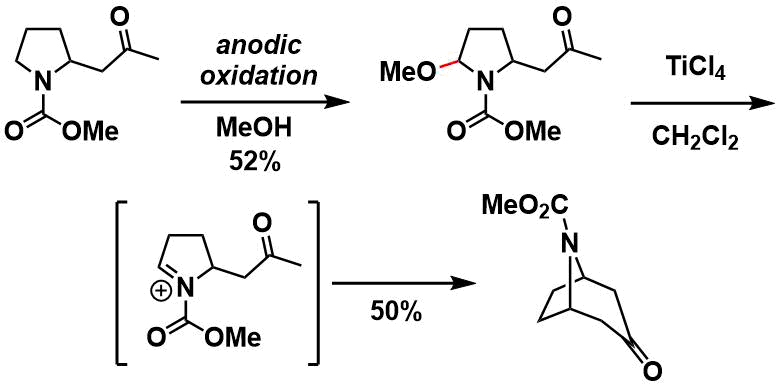
The N,O-acetal product can be carried to subsequent steps to form C-C bonds. Shown below is an example in which it was applied to the synthesis of tropane skeleton.
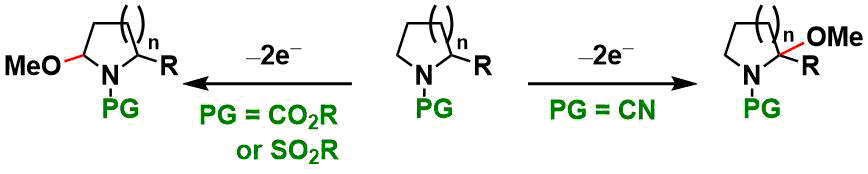
The sterically less hindered side of unsymmetrical substrates is usually oxidized, but the selectivity is reversed for N-cyanoamines.[2] Bicyclocarbamate substrates also react at a more substituted position.[3]
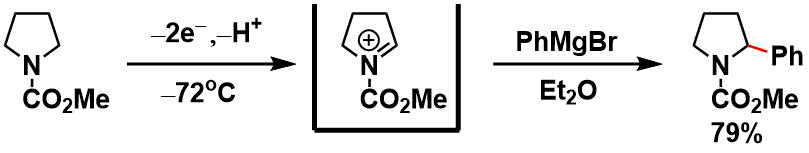
The Shono oxidation under cation pool conditions followed by nucleophilc addition of a Grignard reagent.[4]
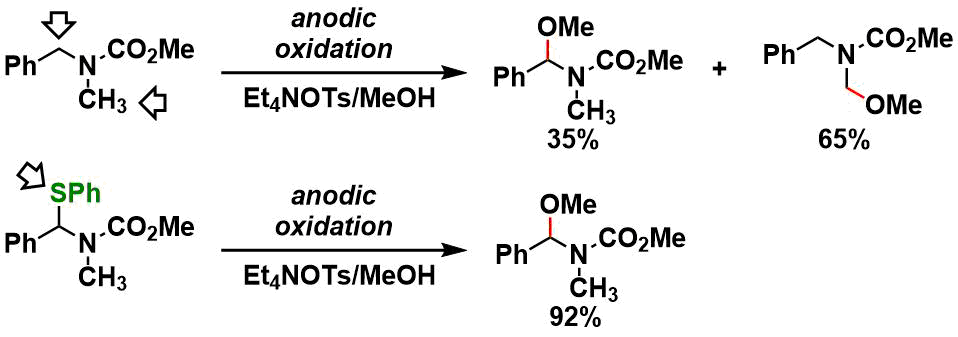
Regioselective oxidation using electroauxiliary[5]: In the example shown below, thiophenyl group works as an electroauxiliary that determines the regioselectivity. Trimethylsilyl (TMS) group is used for the same purpose.
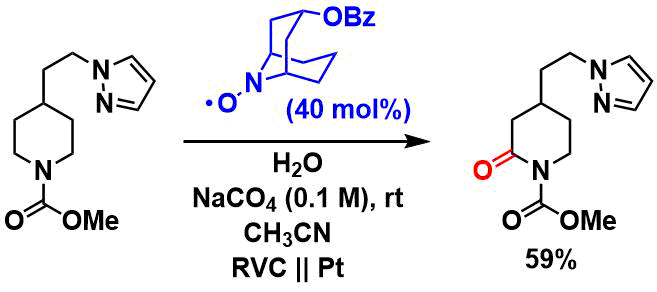
The Shono oxidation with an electrochemical mediator[6]: These systems are highly functional group tolerant.
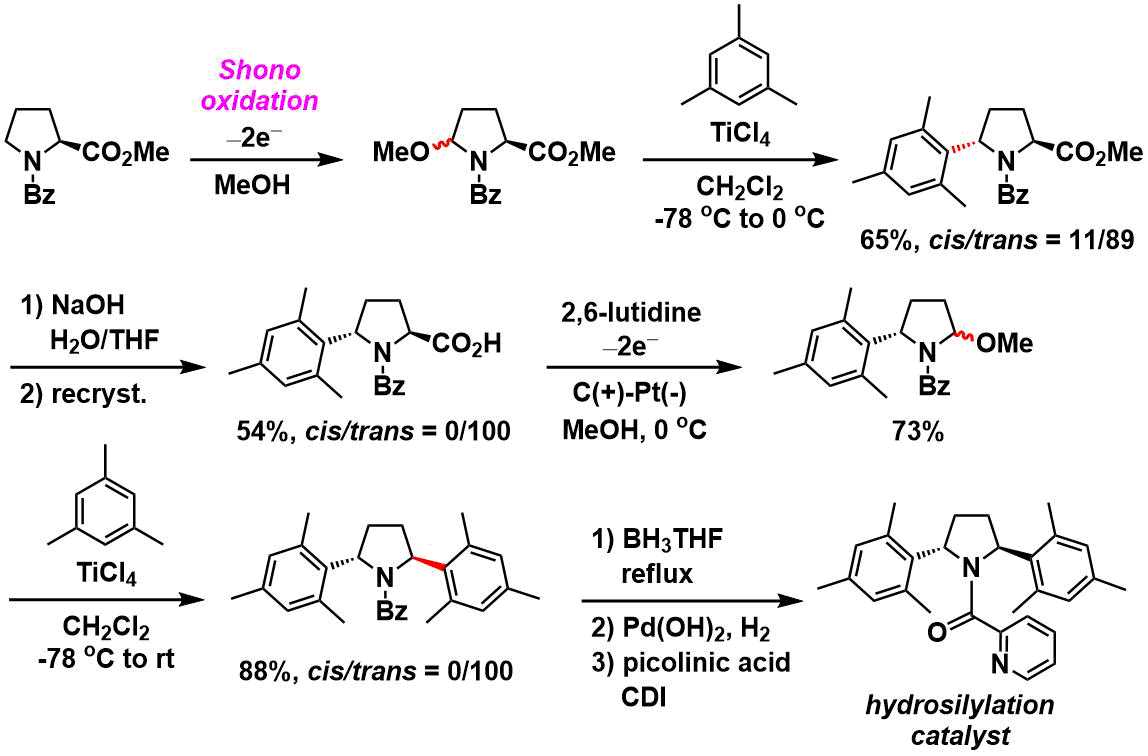
Synthesis of optically active diarylpyrrolidines[7]: A catalyst for asymmetric hydrosilylation was synthesized from proline by way of two Shono oxidation-arylation cycles.
-
Experimental Procedure
-
Experimental Tips
-
References
- Shono, T.; Matsumura, Y.; Tsubata, K. J. Am. Chem. Soc. 1981, 103, 1172. doi:10.1021/ja00395a029
- Libendi, S. S.; Demizu, Y.; Matsumura, Y.; Onomura, O. Tetrahedron 2008, 64, 3935. doi:10.1016/j.tet.2008.02.060
- Onomura, O.; Ishida, Y.; Maki, T.; Minato, D.; Demizu, Y.; Matsumura, Y. Electrochemistry 2006, 74, 645.
- Suga, S.; Okajima, M.; Yoshida, J.-i. Tetrahedron Lett. 2001, 42, 2173. doi:10.1016/S0040-4039(01)00128-9
- Sugawara, M.; Mori, K.; Yoshida, J.-i. Electrochim. Acta 1997, 42, 1995. doi:10.1016/S0013-4686(97)85473-4
- Wang, F.; Rafiee, M.; Stahl, S. S. Angew. Chem. Int. Ed. 2018, 57, 6686. doi:10.1002/anie.201803539
- Onomura, O.; Kirira, P. G.; Tanaka, T.; Tsukada, S.; Matsumura, Y.; Demizu, Y. Tetrahedron 2008, 64, 7498. doi:10.1016/j.tet.2008.06.004
-
Related Reactions
-
Related Books
-
External Links
Electroorganic chemistry(PDF, Baran’s lab)