- Generality
- Unique Reactivity in Photochemistry
- Criteria #3
- Criteria #4
- Criteria #5
-
General Characteristics
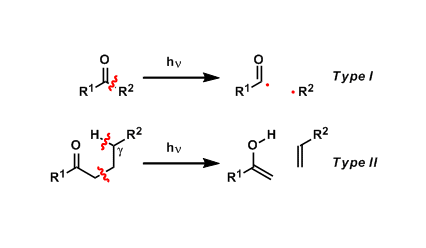
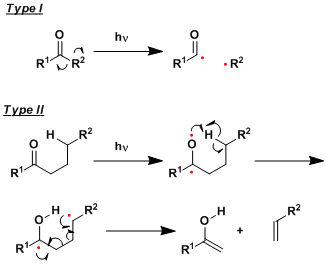
Upon photo-irradiation, carbonyl compounds undergo carbon-carbon bond cleavage reactions via biradical intermediates.
The first type involves the formation of acyl radicals and is called the Norrish type I cleavage. The second type proceeds via γ-hydrogen abstraction and is called the Norrish type II cleavage.
-
General References
- Norrish, R. G. W.; Bamford, C. H. Nature 1936, 138, 1016. doi:10.1038/1381016a0
- Norrish, R. G. W.; Bamford, C. H. Nature 1937, 140, 195. doi:10.1038/140195b0
- Barton, D. H. R.; Charpiot,B.; Ingold, K. U.; Johnston, L. J.; Motherwell, W. B.; Scaiano, J. C.; Stanforth, S. J. Am. Chem. Soc.1985, 107, 3607. DOI: 10.1021/ja00298a034
- Hwu, J. R.; Gilbert, B. A.; Lin, L. C.; Liaw, B. R. J. Chem. Soc. Chem. Commun. 1990, 161. DOI: 10.1039/C39900000161
- Nuss, J. M.; Murphy, M. M. Tetrahedron Lett. 1994, 35, 37. doi:10.1016/0040-4039(94)88156-1
- Henin, F.; M’Boungou-M’Passi, A.; Muzart, J.; Pete, J.-P. Tetrahedron 1994, 50, 2849. doi:10.1016/S0040-4020(01)86998-2
<Reviews>
- Coyle, J. D.; Carless, H. A. J. Chem. Soc. Rev. 1972, 1, 465. DOI: 10.1039/CS9720100465
<Photochemical reactions in total synthesis>
- Hoffmann, N. Chem. Rev. 2008, 108, 1052. DOI: 10.1021/cr0680336
- Bach, T.; Hehn, J. P. Angew. Chem. Int. Ed. 2011, 50, 1000. DOI: 10.1002/anie.201002845
-
Reaction Mechanism
-
Examples
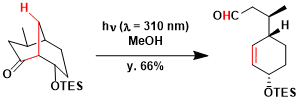
Application to the synthesis of (+)-juvabione.[1]
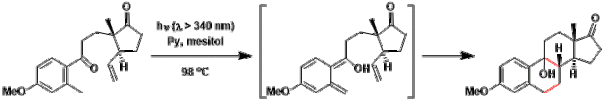
An example of Norrish type II reaction targeted at benzylic C-H bond:[2] The o-quinodimethane intermediate undergoes intramolecular [4+2] cycloaddition.
-
Experimental Procedure
A solution of the ketone (215 mg, 1.01 mmol) in MeOH (100 ml) in Pyrex tubes was degassed by bubbling of argon gas through the solution for 25 min. The solution was irradiated using a Rayonet Photochemical Reactor (Lamp 300 nm) for 90 min at room temperature. Then, the solution was concentrated under reduced pressure and the residue was purified by silica gel column chromatography [hexane-AcOEt (19:1~9:1 v/v)] to afford the aldehyde (149 mg, 69%) as a colorless oil.[1]
-
Experimental Tips
-
References
[1] Itagaki, N.; Iwabuchi, Y. Chem. Commun. 2007, 1175. DOI: 10.1039/B616641E
[2] Quinkert, G.; Stark, H. Angew. Chem. Int. Ed. 1983, 22, 637. DOI: 10.1002/anie.198306373
-
Related Reactions
- [6π]Photocyclization
- Paterno-Büchi Reaction
- [2+2] Photocyclization
- Barton Reaction
- Fries Rearrangment
-
Related Books
[amazonjs asin=”1891389572″ locale=”US” title=”Principles of Molecular Photochemistry: An Introduction”]
-
External Links