- Understanding Reaction Mechanism
- Synthetic Importance
- Criteria #3
- Criteria #4
- Criteria #5
-
General Characteristics
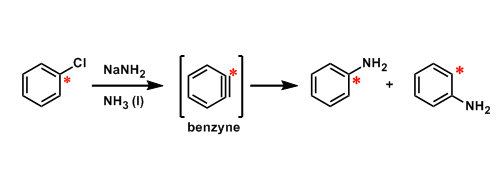
The dehydrogenated form of benzene (the benzene minus two hydrogen atoms) is commonly called benzyne.
There are three regioisomers. o-Benzyne, in which the ortho hydrogens are removed, is most important from synthetic point of view, since it is a strong electorophile and a potent dienophile in the Diels-Alder reaction. p-Benzyne is known as the intermediate of the Masamune-Bergman ene-diyne cyclization.

-
History
In the 1940s, unexpected reactivities of halobenzenes under strongly basic conditions were reported by chemists such as Wittig (1940), Gilman (1945), and Bergstrom (1946). The existence and structure of benzyne, however, was unknown at this point. In 1953, Roberts proposed the reaction mechanism involving benzyne species, based on detailed analysis of the reactions.
“These facts as well as the orientation data for various substituents can be accomodated by an elimination-addition mechanism involving at least transitory existence of an electrically neutral benzyne intermediate.” J. D. Roberts, 1953.
-
General References
<Reviews of aryne>
・Heaney, H. Chem. Rev. 1962, 62, 81. doi:10.1021/cr60216a001
・Pellissier, H.; Santelli, M. Tetrahedron 2003, 59, 701. doi:10.1016/S0040-4020(02)01563-6
・ Wenk, H. H.; Winkler, M.; Sander, W. Angew. Chem. Int. Ed. 2003, 42, 502. DOI: 10.1002/anie.200390151
・ Sanz, R. Org. Prep. Proced. Int. 2008, 40, 215. DOI:10.1080/00304940809458089
・ Kitamura, T. Aust. J. Chem. 2010, 63, 987. doi:10.1071/CH10072
・Tadross, P. M.; Stoltz, B. M. Chem. Rev. 2012, 112, 3550. DOI: 10.1021/cr200478h
・ Gampe, C. M.; Carreira, E. M. Angew. Chem. Int. Ed. 2012, 51, 3766. DOI: 10.1002/anie.201107485
<Reivews of heteroaryne>
・ Goetz, A. E.; Garg, N. K. J. Org. Chem. 2014, 79, 846. doi:10.1021/jo402723e
・ Goetz, A. E.; Shah, T. K.; Garg, N. K. Chem. Commun. 2015, DOI: 10.1039/c4cc06445c
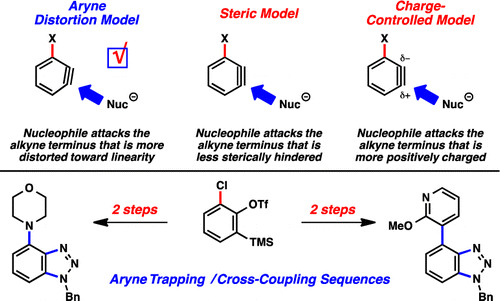
<Aryne distortion models>
・ Bronner, S. M.; Im, G.-Y. J.; Garg, N. K.; Houk, K. N. J. Am. Chem. Soc. 2010, 132, 1267. DOI: 10.1021/ja9098643
・ Im, G-Y. J.; Bronner, S. M.; Goetz, A. E.; Paton, E. S.; Cheong, P. H.-Y.; Houk, K. N.; Garg, N. K. J. Am. Chem. Soc. 2010, 132, 17933. DOI: 10.1021/ja1086485
・Goetz, A. E.; Bronner, S. M.; Cisneros, J. D.; Melamed, J. M.; Paton, E. S.; Houk, K. N.; Garg, N. K. Angew. Chem. Int. Ed. 2012, 51, 2758. DOI: 10.1002/anie.201108863
・Goetz, A. E.; Garg, N. K. Nat. Chem. 2013, 5, 54. doi:10.1038/nchem.1504
・Medina, J. M.; Mackey, J. L.; Garg, N, K.; Houk, K. N. J. Am. Chem. Soc. 2014, 136, 15798. DOI: 10.1021/ja5099935
-
Reaction Mechanism
Because the triple bond of o-benzyne is distorted heavily from the most stable straight angle, it is a very unstable and reactive species.
The length of benzyne’s triple bond (1.24 Å) is between that of typical double bonds (1.34 Å, ethylene) and triple bonds (1.20 Å, acetylene).
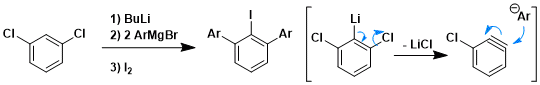
Benzyne is strongly electrophilic in solution. For example, the reaction shown in the top of this page is known to involve a benzyne intermediate and is not nucleophilic aromatic substitution. This mechanisum is supported by carbon isotope labelling (of red asterisk positions).
The regioselectivity of nucleophilic attack on benzynes is influenced by substituents. Recently, the aryne distortion model has been proposed to explain the regioselectivity.
-
Examples
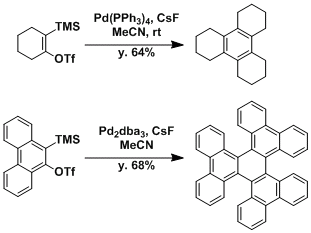
Cyclotrimerization of benzynes.
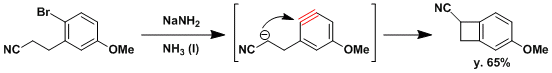
Intramolecular nucleophilic attack to form a 4-membered ring.[1]
Synthesis of a terphenyl derivative.
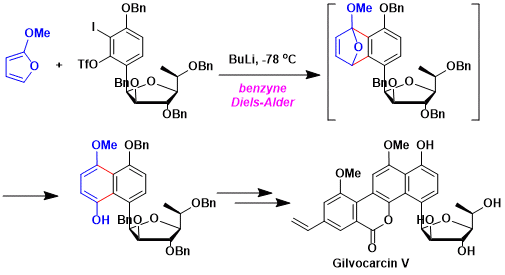
Synthesis of gilvocarcin compounds.[2]
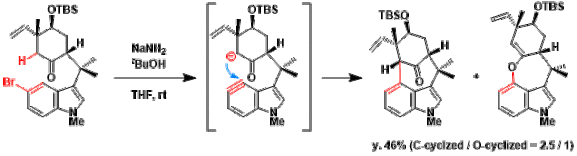
Total synthesis utilizing a key step involving indolyne species.[3]
-
Experimental Procedure
Acylalkylation via benzyne intermediate.[4]
An oven-dried 500-mL, three-necked, round-bottomed flask is equipped with a magnetic stir bar, and cesium fluoride (19.7 g, 130 mmol, 2.5 equiv) is added. The flask is fitted with a reflux condenser in the middle neck, an adaptor equipped with a thermometer in one of the side necks, and a septum in the other side neck. The condenser is equipped with a vacuum adaptor connected to a Schlenk line (nitrogen/vacuum manifold). The connected glassware is evacuated under high vacuum (0.025 mmHg) and carefully back-filled with nitrogen. This procedure is repeated twice to secure an oxygen-free atmosphere in the flask. Dry MeCN (260 mL) is added to the flask via a syringe through the septum. While stirring the reaction mixture, methyl acetoacetate (5.60 mL, 6.01 g, 51.8 mmol, 1.00 equiv) and 2-(trimethylsilyl)phenyl trifluoromethanesulfonate (15.7 mL, 19.3 g, 64.7 mmol, 1.25 equiv) are added through the septum via syringes. The flask is submerged in an oil bath (100 °C) and the reaction mixture is heated to reflux (internal temperature: 78-81 °C, reached after 15 min). The mixture is stirred at reflux temperature for 40 min. Initially, the reaction mixture is a white opaque suspension, but during heating it changes color to yellow and upon reflux the color changes to orange. As the reaction progresses the opaque solution becomes transparent and the color changes back to yellow. Throughout the entire time a white precipitate is present in the flask. The reaction flask is removed from the oil bath and allowed to cool to ambient temperature (23 °C) over the course of 1 h. The flask is disconnected from the condenser and the nitrogen inlet, and the reaction mixture is diluted with saturated aqueous NaCl solution (200 mL). This mixture is carefully poured into a 1-L separatory funnel. After the layers are separated, the aqueous layer is extracted with Et2O (3 × 200 mL). The combined organic layers are dried over anhydrous Na2SO4 and filtered. Concentration of the dried organic layers is effected by rotary evaporation (35 °C, 45 mmHg) which affords an orange, viscous oil. Partial purification is achieved by flash chromatography (4.5 × 24 cm, 170 g silica gel) using a gradient of hexanes to 40% Et2O in hexanes. The crude product is loaded on the column with benzene (20 mL). Fraction collection (50 mL fractions) is begun as the crude product is eluted first with 100 mL of hexanes, followed by 1500 mL of 9:1 hexanes:Et2O, 2000 mL of 4:1 hexanes:Et2O, 1000 mL of 7:3 hexanes:Et2O and finally 750 mL of 3:2 hexanes:Et2O. Fractions 38-85 are concentrated by rotary evaporation (30-35 °C, 45 mmHg) to afford 7.96 g (80%) of a slightly yellow solid. The partially purified product is further purified by bulb-to-bulb distillation at 124-130 °C (0.75 mmHg) which affords 6.63 g (67%) of the title compound as an off-white, crystalline solid.
-
Experimental Tips
-
References
[1] Agami, C.; Couty, F.; Poursolis, M.; Vaissermann, J. Tetrahedron 1992, 48, 431. doi:10.1016/S0040-4020(01)89005-0
[2] Hosoya, T.; Takashiro, E.; Matsumoto, T.; Suzuki, K. J. Am. Chem. Soc. 1994, 116, 1004. doi:10.1021/ja00082a023
[3] Huters, A. D.; Quasdorf, K. W.; Styduhar, E. D.; Garg, N. K. J. Am. Chem. Soc. 2011, 133, 15797. doi:10.1021/ja206538k
[4] Ebner, D. C.; Tambar, U. K.; Stoltz. B. M. Org. Synth. 2009, 86, 161. [website]
-
Related Reactions
-
Related Books
[amazonjs asin=”3718600404″ locale=”US” title=”Azosulfones: Versatile Precursors for Aryl Radicals, Aryl Cations, Aryl Anions, Carbenes and Benzynes”]
[amazonjs asin=”1468472925″ locale=”US” title=”Carbenes nitrenes and arynes (Studies in Modern Chemistry)”]
-
External Links
- Aryne – Wikipedia
- Benzyne (Stoltz group, PDF)