- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
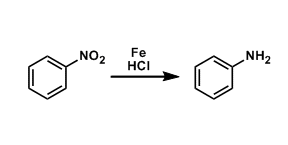
The reduction of aromatic nitro group using iron powder and HCl is called the Bechamp reduction. Besides these classical conditions, other reagents such as tin(II) chloride are also used.
-
General References
- Bechamp, A. J. Ann. Chim. Phys. 1854, 42, 186.
- Org. React.1944, 2, 428.
-
History
This reaction was discovered by the French chemist Antoine Béchamp in 1854. Béchamp is also known for developing electrophilic aromatic substitution of phenols and anilines using arsenic acid.
-
Reaction Mechanism
-
Examples
-
Experimental Procedure
Reduction of 4-Nitroacetophenone. (PDF)
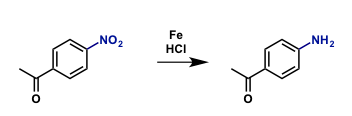 70 mL of ethanol are placed in a 250 mL–three-necked round bottom flask equipped with a reflux condenser, a dropping funnel, a glass stopper and a magnetic stirring bar. 3.30 g (20.0 mmol) of 4Nitroacetophenone and 4.19 g (75.0 mmol) of iron powder are added. The mixture is heated to 60°C and 15 mL of concentrated hydrochloric acid are added dropwise within 30 min. Subsequently, the reaction mixture is refluxed for 1 hour until the iron powder is mostly dissolved. The reaction mixture is cooled down and poured into a 1000 mL Duran flask containing 200 mL of water and a magnetic stirring bar. Stirring is commenced and the solution is neutralized with diluted sodium hydroxide solution (c = 2 mol/L) (formation of iron hydroxide sludge!!). The pH is measured by means of a pH-paper. Subsequently, 150 mL of ethyl acetate are added. Extraction of the organic compounds is achieved by stirring the mixture for 15 min. The stirring process is stopped. Wait until the two phases have separated. Decant most of the upper organic layer into the beaker. The residual extraction liquids are poured into a 500 ml separating funnel. Wait until the two layers have separated. Gently swing the separating funnel in upright position so that the solid iron hydroxide settles out. (Do not shake vigorously, since an emulsion can be formed). Separate the two layers. The combined organic layers are dried over anhydrous sodium sulfate. Subsequently, the drying agent is sucked off. The organic layer is transferred into a 250 mL round bottom flask which had been weighed. The organic solvent is distilled off under vacuum. Based on the reaction scheme calculate the percent yield of the crude product. For recrystallisation the as-prepared raw material is boiled in 150 mL of water. The hot solution is filtered under suction through a Büchner funnel. The pure product crystallizes on cooling, is filtered off and dried in a desiccator.
70 mL of ethanol are placed in a 250 mL–three-necked round bottom flask equipped with a reflux condenser, a dropping funnel, a glass stopper and a magnetic stirring bar. 3.30 g (20.0 mmol) of 4Nitroacetophenone and 4.19 g (75.0 mmol) of iron powder are added. The mixture is heated to 60°C and 15 mL of concentrated hydrochloric acid are added dropwise within 30 min. Subsequently, the reaction mixture is refluxed for 1 hour until the iron powder is mostly dissolved. The reaction mixture is cooled down and poured into a 1000 mL Duran flask containing 200 mL of water and a magnetic stirring bar. Stirring is commenced and the solution is neutralized with diluted sodium hydroxide solution (c = 2 mol/L) (formation of iron hydroxide sludge!!). The pH is measured by means of a pH-paper. Subsequently, 150 mL of ethyl acetate are added. Extraction of the organic compounds is achieved by stirring the mixture for 15 min. The stirring process is stopped. Wait until the two phases have separated. Decant most of the upper organic layer into the beaker. The residual extraction liquids are poured into a 500 ml separating funnel. Wait until the two layers have separated. Gently swing the separating funnel in upright position so that the solid iron hydroxide settles out. (Do not shake vigorously, since an emulsion can be formed). Separate the two layers. The combined organic layers are dried over anhydrous sodium sulfate. Subsequently, the drying agent is sucked off. The organic layer is transferred into a 250 mL round bottom flask which had been weighed. The organic solvent is distilled off under vacuum. Based on the reaction scheme calculate the percent yield of the crude product. For recrystallisation the as-prepared raw material is boiled in 150 mL of water. The hot solution is filtered under suction through a Büchner funnel. The pure product crystallizes on cooling, is filtered off and dried in a desiccator.
-
Experimental Tips
-
References
-
Related Reactions
-
Related Books
[amazonjs asin=”B009WOKMQW” locale=”US” title=”Oxidation and reduction in organic chemistry from the standpoint of potential differences; the system hydroquinone and quinone”]
-
External Links
- Bechamp Reduction – Wikipedia
- Reduction of Nitro Compounds – Wikipedia