- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #5
-
General Characteristics
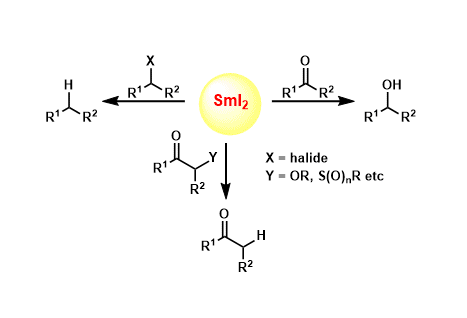
Samarium(II) iodide (SmI2) is a mild one-electron reducing agent used for various reductive reactions of carbonyl compounds, organic halides, and electron deficient olefins.
Additional applications of SmI2 include pinalol coupling, alkyl-carbonyl coupling, deoxygenation, the Reformatsky reaction, and free-radical mediated cyclization.
Recent studies have shed light on the effect of additives on the reactivity of SmI2.
-
General References
- Girard, P.; Namy, J. L.; Kagan, H. B. J. Am. Chem. Soc. 1980, 102, 2693. DOI: 10.1021/ja00528a029
<Reviews>
- Molander, G. A. Harris, C. R. Chem. Rev. 1996, 96, 307. DOI: 10.1021/cr950019y
- Molander, G. A. Org. React.2004, DOI: 10.1002/0471264180.or046.03
- Edmonds, D. J.; Johnston, D.; Procter, D. J. Chem. Rev.2004, 104, 3371. DOI: 10.1021/cr030017a
- Nicolaou, K. C.; Ellery, S. P.; Chen, J. S. Angew. Chem. Int. Ed.2009, 48, 7140. doi: 10.1002/anie.200902151
- Szostak, M.; Procter, D. J. Angew. Chem. Int. Ed.2012, 51, 9238. DOI: 10.1002/anie.201201065
- Szostak, M.; Spain, M.; Procter, D. J. Chem. Soc. Rev.2013, 42, 9155. DOI: 10.1039/C3CS60223K
-
Reaction Mechanism
The reactivity of SmI2 is known to vary depending on the additive. In particular, Lewis bases such as HMPA and Et3N and proton sources like BuOH and H2O improve the chemoselectivity and reduction potential.
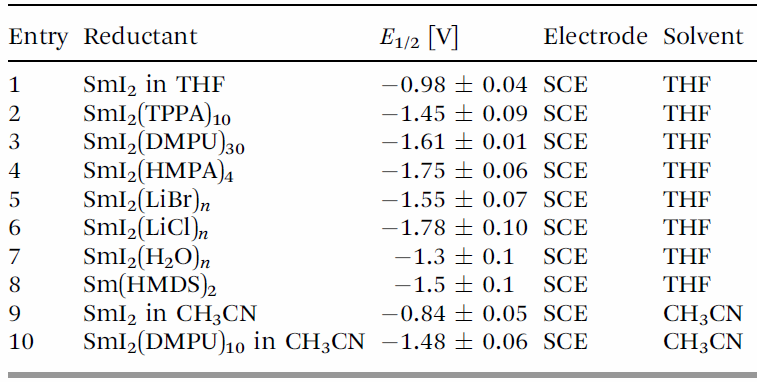
(The table is extracted from Chem. Soc. Rev. 2013, 42, 9155.)
-
Examples
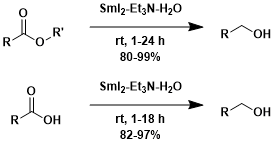
The SmI2-amine-H2O combination is capable of reducing esters and carboxylic acids.[1]
Total synthesis of (+)-pleuromutilin[2]: SmI2 provided a highly efficient means of ester reduction, where the yield with LiAlH4 was only 21%.
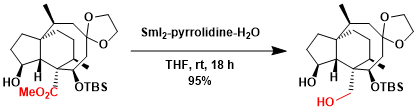
SmI2 is a valuable reagent for mild deprotection of Ts group.[3]
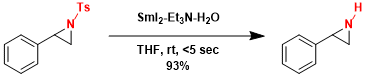
Total synthesis of maoecrystal Z[4]: SmBr2 led to a better result than SmI2 in this case.
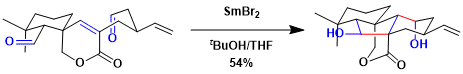
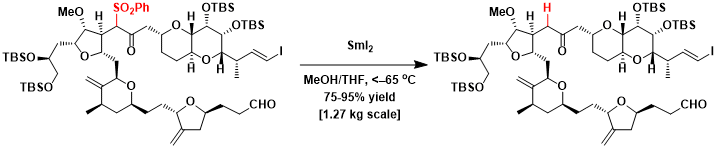
Kilo-scale synthesis of chemotherapy drug Halaven[5]: The highly selective reduction was achieved by careful optimization of reaction conditions.
Total synthesis of atractyligenin[6]: The directing effect of the neighboring axial ester group was utilized to achieve the highly stereoselective ketone reduction.
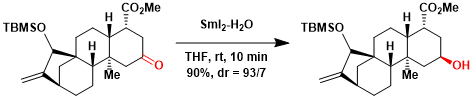
The deprotection of allyl ethers is another application.[7]
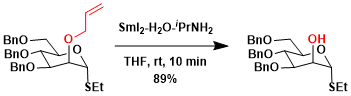
-
Experimental Procedure
Preparation of SmI2.[8]
![]()
An oven-dried 100 mL round-bottomed flask equipped with a Teflon-coated magnetic stir bar and a septum was placed under vacuum and evacuated/backfilled with argon three times. In air, 1.65 g (11.0 mmol) of samarium metal and 1.55 g (5.5 mmol) of freshly washed 1,2-diiodoethane (see, below) was weighed out and added to the reaction flask. The flask was sealed with a septum, evacuated/backfilled with argon three times, and stirring was started at medium speed. 55 mL of commercial THF was added using 60 mL syringe. The flask was evacuated/backfilled with argon three times (this process removes ethylene formed during the insertion of Sm into 1,2-diiosoethane), the argon line was removed, and the septum was sealed with Parafilm. After stirring overnight, the stirring was turned off, and the solution of SmI2 was allowed to settle for 30 min and titrated.
Purification of diiodoethane.[8]
1,2-Diiodoethane (20 g) was dissolved in approximately 400 mL of diethyl ether, and the organic layer was washed with aqueous saturated sodium thiosulfate solution (5 x 100 mL) and water (1 x 100 mL), dried over sodium sulfate, and concentrated to give a white solid. The flask containing 1,2-diiodoethane was wrapped in aluminum foil and placed under high-vacuum for 30 min.
-
Experimental Tips
1) SmI2 is air-sensitive, thus it needs to be stored in well-degassed solvents and under argon atmosphere.
2) It has been reported that the quality of SmI2 is affected most by the quality of samarium metal, rather than water and oxygen.[8]
The color of SmI2 solutions looks like the following:

(a) THF solution (blue), (b) H2O solution (wine red), (c) after being consumed by the reaction (white/yellow)
-
References
[1] (a) Szostak, M.; Spain, M.; Procter, D. J. Chem. Commun. 2011, 47, 10254. DOI: 10.1039/C1CC14014K (b) Szostak, M.; Spain, M.; Procter, D. J. Org. Lett. 2012, 14, 840. DOI: 10.1021/ol203361k [2] Fazakerley, N. J.; Helm, M. D.; Procter, D. J. Chem. Eur. J. 2013, 19, 6718. DOI: 10.1002/chem.201300968 [3] Ankner, T.; Hilmersson, G. Org. Lett. 2009, 11, 503. DOI: 10.1021/ol802243d [4] Cha, J. Y.; Yeoman, J. T. S.; Reisman, S. E. J. Am. Chem. Soc. 2011, 133, 14964. DOI: 10.1021/ja2073356 [5] Eisai Inc, Synlett 2013, 333. DOI: 10.1055/s-0032-1318026 [6] Singh, A. K.; Bakshi, R. K.; Corey, E. J. J. Am. Chem. Soc. 1987, 109, 6187. DOI: 10.1021/ja00254a051 [7] Dahlen, A.; Sundgren, A.; Lahmann, M.; Oscarson, S.; Hilmersson, G. Org. Lett. 2003, 5, 4085. doi:10.1021/ol0354831 [8] Szostak, M.; Spain, M.; Procter, D. J. J. Org. Chem. 2012, 77, 3049. DOI: 10.1021/jo300135v [9] Szostak, M.; Spain, M.: Procter, D. J. Nature Protocols, 2012, 7, 970. DOI:10.1038/nprot.2012.034
-
Related Reactions
-
Related Books
[amazonjs asin=”1847551106″ locale=”US” title=”Organic Synthesis using Samarium Diiodide: A Practical Guide”]
-
External Links
- Samarium (II) Iodide – Wikipedia
- Reductions with samarium(II) iodide – Wikipedia
- Samarium(II) Iodide in Organic Synthesis (PDF, MacMillan’s group)
- Some Synthetic Applications of Samarium (II) Iodide (PDF)
- SAMARIUM DIIODIDE: DISCOVERY AND APPLICATIONS (PDF, Dong group)
- Samarium(II) Iodide (PDF, Baran’s group)
- Procter Group
- Samarium (low valent) (organic-chemistry.org)
- Preparation and Use of Samarium Diiodide (SmI2) in Organic Synthesis (JOVE)