- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
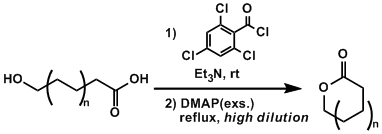
In the method developed by Yamaguchi, hydroxycarboxylic acids (“seco-acids”) are converted into reactive acid anhydrides using 2,4,6-trichlorobenzoyl chloride and the intramolecular esterification is promoted by DMAP under high dilution conditions. This reaction often requires refluxing temperatures depending on the size of the ring being formed.
-
General References
・Inanaga, J.; Hirata, K.; Saeki, H.; Katsuki, T.; Yamaguchi, M. Bull. Chem. Soc. Jpn. 1979, 52, 1989. DOI: 10.1246/bcsj.52.1989
・Kawanami, Y.; Dainobu, Y.; Inanaga, J.; Katsuki, T.; Yamaguchi, M. Bull. Chem. Soc. Jpn. 1981, 54, 943. DOI: 10.1246/bcsj.54.943
-
Reaction Mechanism
The acid anhydrides react regioselectively due to the steric hindrance by the ortho-substitution on the benzoyl group (Ref: Org. Lett.2006, 8, 47.).
-
Examples
The Yamaguchi conditions are compatible with a wide range of substrates and are used frequently even today. An example shown below is the esterification between two large fragments in the total synthesis of cryptophycin-24.[1]
-
Experimental Procedure
-
Experimental Tips
-
References
[1] Eggen, M.; Mossman, C. J. et al. J. Org. Chem. 2000, 65, 7792. DOI: 10.1021/jo000767+
-
Related Books

![yamaguchi_macrolactone_2[1]](https://assets.en.chem-station.com/uploads/2014/08/yamaguchi_macrolactone_21.gif)
![yamaguchi_macrolactone_3[1]](https://assets.en.chem-station.com/uploads/2014/08/yamaguchi_macrolactone_31.gif)