- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
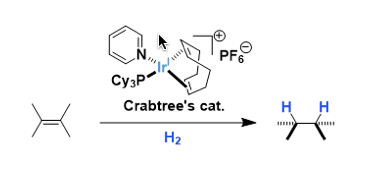
[Ir(cod)(PCy3)(Py)]PF6 is an easily handleable cationic iridium complex used as a homogeneous hydrogenation catalyst. The complex was developed by Robert Crabtree of Yale University.
It is active enough to catalyze hydrogenation of tetrasubstituted olefins, which is generally difficult with other catalysts (such as the Wilkinson’s catalyst). The hydrogenation can be highly selective in the presence of coordinating functional groups.
-
General References
- Crabtree, R. H. Acc. Chem. Res. 1979, 12, 331. doi:10.1021/ar50141a005
- Crabtree, R. H. Davis, M. W. J. Org. Chem. 1986, 51, 2655. DOI: 10.1021/jo00364a007
<Reviews of Directed Hydrogenation>
- Brown, J. M. Angew. Chem. Int. Ed. 1987, 26, 190. doi:10.1002/anie.198701901
-
Reaction Mechanism
-
Examples
The synthesis of (-)-calyciphylline N[1]: The highly active catalyst containing a more loosely coordinating BARF anion[2] was used for hydrogenation of the tetrasubstituted olefin.

An example of stereoselective hydrogenation directed by neighboring functional groups.

-
Experimental Procedure
-
Experimental Tips
-
References
[1] Shvartsbart, A.; Smith, A. B., III J. Am. Chem. Soc. 2014, 136, 870. DOI: 10.1021/ja411539w
[2] Wustenberg, B.; Pfaltz, A. Adv. Synth. Catal. 2008, 350, 174. DOI: 10.1002/adsc.200700438
-
Related Reactions
-
Related Books
[amazonjs asin=”3527311610″ locale=”US” title=”Handbook of Homogeneous Hydrogenation (3 Volumes)”]
[amazonjs asin=”9048143233″ locale=”US” title=”Homogeneous Hydrogenation (Catalysis by Metal Complexes)”]
-
External Links