Khusnutdinova, J. R.; Ben-David, Y.; Milstein, D. J. Am. Chem. Soc. 136, 2014. 2998.
DOI: 10.1021/ja500026m
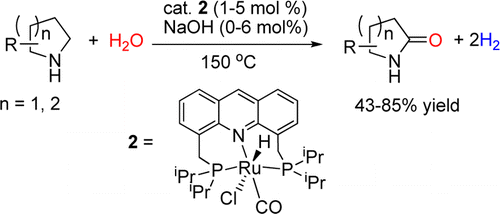
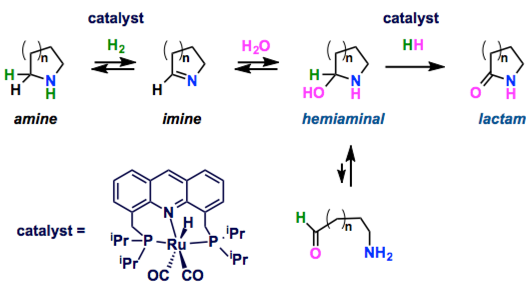
Direct conversion of cyclic amines to lactams utilizing water as the only reagent is catalyzed by pincer complex. In contrast to previously known methods of amine-to-amide conversion, this reaction occurs in the absence of oxidants and is accompanied by liberation of H2, with water serving as a source of oxygen atom. Formation of a cyclic hemiaminal intermediate plays a key role in enabling such reactivity. This represents an unprecedented, conceptually new type of amide formation reaction directly from amines and water under oxidant-free conditions.
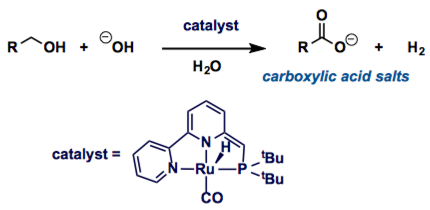
Cyclic and acyclic amides are common components of naturally occurring compounds, biologically active compounds pharmaceuticals. Recently, Milstein group reported the Ru complex-catalyzed the direct dehydrogenation of alcohols in water to give carboxylic acid salts selectively with a stochiometric amount of base.[1]
In this report, they show the direct conversion of amines to amides via oxygenation of an α-methylene. The synthesis of lactams proceeded via a Ru complex-catalyzed dehydrogenation of cyclic amines in water under oxidant free condition. Milstein proposed that the lactams formed via cyclic hemiaminal intermediates which are entropically favored against the elimination of amine. Then, the further dehydrogenation to the lactams would occur.
-
References
[1]“Catalytic transformation of alcohols to carboxylic acid salts and H2 using water as the oxygen atom source”
Balaraman, E.; Khaskin, E.; Leitus, G.; Milstein, D. Nat. Chem. 5, 2013. 122. DOI: 10.1038/nchem.1536
The oxidation of alcohols to carboxylic acids is an important industrial reaction used in the synthesis of bulk and fine chemicals. Most current processes are performed by making use of either stoichiometric amounts of toxic oxidizing agents or the use of pressurized dioxygen. Here, we describe an alternative dehydrogenative pathway effected by water and base with the concomitant generation of hydrogen gas. A homogeneous ruthenium complex catalyses the transformation of primary alcohols to carboxylic acid salts at low catalyst loadings (0.2 mol%) in basic aqueous solution. A consequence of this finding could be a safer and cleaner process for the synthesis of carboxylic acids and their derivatives at both laboratory and industrial scales.
-
Related Links
Milstein Lab home Page