Chong, C.-C.; Hirao, H.; Kinjo, R. Angew. Chem. Int. Ed. 53, 2014. Early View
DOI: 10.1002/anie.201400099
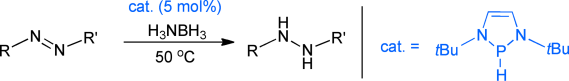
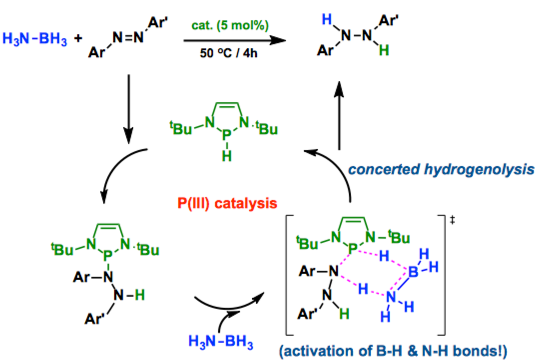
1,3,2-diazaphospholenes catalyze metal-free transfer hydrogenation of a NN double bond using ammonia–borane under mild reaction conditions, thus allowing access to various hydrazine derivatives. Kinetic and computational studies revealed that the rate-determining step involves simultaneous breakage of the BH and NH bonds of ammonia–borane. The reaction is therefore viewed as a concerted type of hydrogenolysis.
Recent breakthroughs in frontier inorganic chemistry have demonstrated that various main-group compounds are able to activate small molecules featuring the strong bonds such as H-H, N-H, C-H. Thus, p-block elements can be utilized as mimics for transition metals center. Although there are a few papers describing the activation of B-H and N-H bonds of ammonia borane by main group compounds, their applications in catalysis remain extremely challenging.
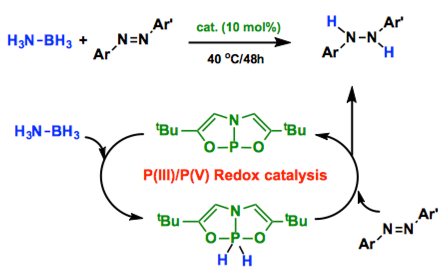
Only Kinjo group developed a novel transfer hydrogenation system using N-heterocyclic phosphane as a catalyst and ammonia-borane as the source of hydrogen. By stoichiometric reaction, kinetic experiments, and theoretical calculation, they revealed the unique catalytic cycle involves (1) the addition of the P-H bond to the N=N double bond leading to phosphinohydrazines, and (2) the hydrogenolysis of the exocyclic P-Nbond via concerted hydrogen transfer from ammonia-borane. Importantly, the catalyst maintains the PIII oxidation state throughout the whole catalytic cycle, which is totally different from the P(III)/P(V) redox cycle discovered by Radosevich group.[1-2]
-
References
[1] “Main Group Redox Catalysis: Reversible P(III)/P(V) Redox Cycling at a Phosphorus Platform”
Dunn, N. L.; Ha, M.; Radosevich, A. T. J. Am. Chem. Soc. 2012, 134, 11330 – 11333. DOI: 10.1021/ja302963p
[2] “Main Group Bonsai Phosphorus”
Coyle, E. E.; O’Brien, C. J. Nat. Chem. 2012, 4, 779 – 780. DOI: 10.1038/nchem.1470
A planar, trivalent phosphorus compound is shown to undergo reversible two-electron redox cycling (PIII/PV) enabling its use as catalyst for a transfer hydrogenation reaction. The trivalent phosphorus compound activates ammonia-borane to furnish a 10-P-5 dihydridophosphorane, which in turn is shown to transfer hydrogen cleanly to azobenzene, yielding diphenylhydrazine and regenerating the initial trivalent phosphorus species. This result constitutes a rare example of two electron redox catalysis at a main group compound and suggests broader potential for this nonmetal platform to support bond-modifying redox catalysis
-
Related Links
Kinjo Lab home Page
Radosevich Lab home Page