Asadirad, A. M.; Boutault, S.; Erno, Z.; Branda, N. R. J. Am. Chem. Soc. 2014. ASAP
DOI: 10.1021/ja500496n
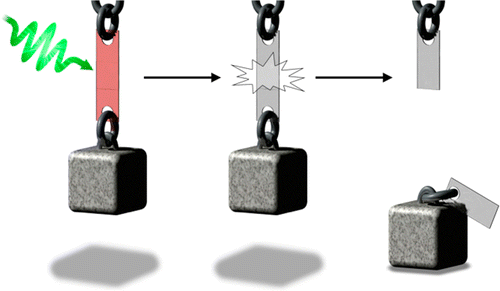
A thermally remendable polymer was synthesized by the Diels–Alder reaction between dithienylfuran and maleimide monomers to generate a photoresponsive diarylethene. UV light (312 nm) and visible light (>435 nm) “gate” the reversibility of the Diels–Alder reaction and turn the self-healing properties of the polymer “off” and “on”, respectively. After exposure to UV light, the strength of the polymer as an adhesive is enhanced. Visible light weakens the adhesive.
Prof. Baranda and coworkers, department of chemistry, Simon Fraser University, reported the synthesis of thermally remendable polymer. Baranda group program lies at the interface of organic chemistry and materials science with a focus on designing and synthesizing molecular switches.
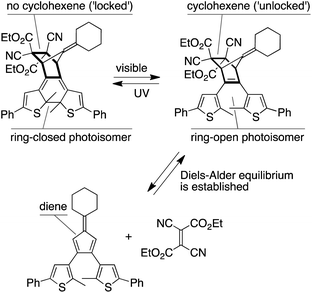
They have recently demonstrated that they can “gate’” reverse Diels−Alder reactions in small molecules by integrating the diene component into a photoresponsive hexatriene.[1] In this paper, they show that this approach works equally well to control the properties of linear polymers made up of furan rings as the dienes and maleimides as the dienophiles. As a results, they have demonstrated how two wavelengths of light can be used to regulate the adhesive properties of a photoresponsive polymer by inducing ring-closing and ring- opening reactions of the polymers’ building blocks.
-
References
[1] “Using light and a molecular switch to ‘lock’ and ‘unlock’ the Diels–Alder reaction”
Erno, Z.; Asadirad, A. M.; Lemieux, V. Org. Biomol. Chem. 2012 ,10, 2787.
Light is used to ‘gate’ the Diels–Alder reaction using a photoresponsive dithienylfuran backbone and turn the reversibility of the Diels–Alder reaction ‘off’ and ‘on’ at 100 °C. These features make the reported system an excellent candidate for developing the next generation of self-healing polymers and photothermal drug delivery vehicles.
- Related Books
[amazonjs asin=”3527313656″ locale=”US” title=”Molecular Switches”]
- Related Links
Branda Research Group