Protchenko, A. V.; Dange, D.; Harmer, J. R.; Tang, C. Y.; Schwarz, A. D.; Kelly, M. J.; Phillips, N.; Tirfoin, R.; Birjkumar, K. H.; Jones, C.; Kaltsoyannis, N.; Mountford. P; Aldridge, S. Nat. Chem., 2014. ASAP
DOI: 10.1038/nchem.1870
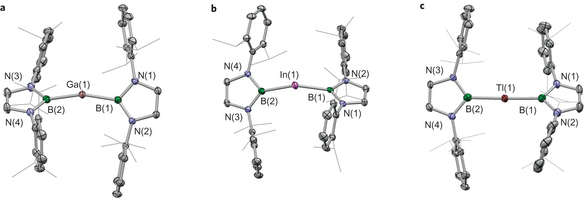
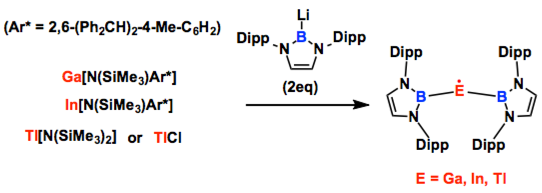
The chemistry of the Group 13 metals is dominated by the +1 and +3 oxidation states, and simple monomeric M(II) species are typically short-lived, highly reactive species. Here we report the first thermally robust monomeric MX2 radicals of gallium, indium and thallium. By making use of sterically demanding boryl substituents, compounds of the type M(II)(boryl)2 (M = Ga, In, Tl) can be synthesized. These decompose above 130 oC and are amenable to structural characterization in the solid state by X-ray crystallography. Electron paramagnetic resonance and computational studies reveal a dominant metal-centred character for all three radicals (>70% spin density at the metal). M(II) species have been invoked as key short-lived intermediates in well-known electron-transfer processes; consistently, the chemical behaviour of these novel isolated species reveals facile one-electron shuttling processes at the metal centre.
Similar to carbene chemistry which has been extensively studied by Arduengo and Bertrand groups and widely used in several fields by many chemists all over the world, the boryl lithium originally developed by Nozaki group has recently been recognized an efficient motif to cultivate new fields. Because the boryl lithium seems readily available synthetically, the number of applications with boryl group has increased in recent papers. Here, Aldridge group reported the unique property of boryl group.
Thus, it can stabilize the radical species of heavier group 13 elements that usually cannot exist or are persistent under normal conditions. They completed the fully characterization of EPR spectra as well as molecular structure determination by X-ray for all products. Wondering if they could isolate Al and B analogues by using same way? The relevant precursors were also reported by Nozaki and Yamashita groups [1]
-
References
[1] “Syntheses and properties of triborane(5)s possessing bulky diamino substituents on terminal boron atoms”
Hayashi, Y.; Segawa, Y.; Yamashita, M.; Nozaki, K. Chem. Commun. 2011, 47, 5888 – 5890.DOI: 10.1039/c1cc11334h
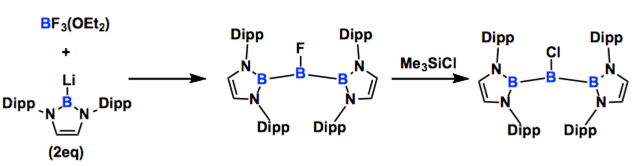
The present communication reports the chemistry of three linear triborane(5) compounds possessing bulky diamino substituents on the terminal boron atoms. Fluorotriborane was synthesized by a reaction of boryllithium and BF3OEt2. Halogen exchange reaction of Fluorotriborane took place by a treatment with ClSiMe3 to give the corresponding chlorotriborane(5). Addition of silver tetraarylborate to chlorotriborane(5) in ether afforded a hydroxylated triborane(5) compound probably via an unstable cationic boron intermediate.
-
Related Links
Aldridge Lab home Page
Nozaki Lab home Page
Yamashita Lab home Page