Jana, A.; Huch, V.; Repisky, M.; Berger, R. J. F.; Scheschkewitz, D. Angew. Chem. Int. Ed., 2014. Early View
DOI: 10.1002/anie.201310475
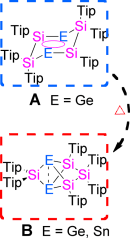
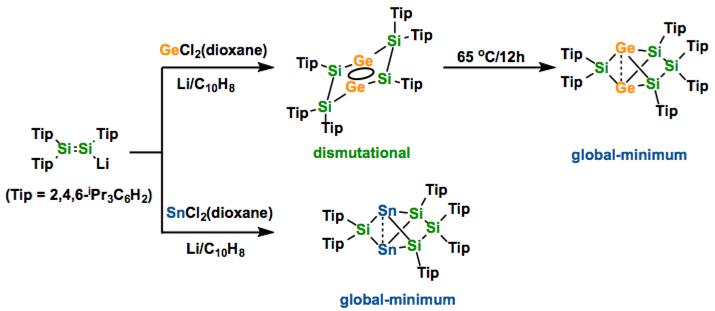
Aromatic species with heavier Group 14 elements show remarkable differences in terms of stability, structure, and reactivity. Herein we report our experimental and theoretical investigations regarding isomers of germanium- and tincontaining benzene analogues E2Si4R6 (E=Ge, Sn). The germanium-substituted dismutational isomer with a tricyclic six-membered scaffold is isolable, but unlike the homonuclear Si6 analogue slowly rearranges even at room temperature to give the propellane-type global minimum isomer. In case of E=Sn the dismutational isomer may be an intermediate on the pathway to the propellane-type species obtained, but cannot be detected even at low temperature. Unprecedentedly large chemical shift anisotropies in the 29Si NMR spectra that increase from the Si6 species through Ge2Si4 to Sn2Si4 are rationalized by progressively larger paramagnetic-term contributions to the chemical shift tensor as a result of diminishing HOMO–LUMO gaps, which are also reflected in the absorption spectra, as well as by appearance and symmetry of these frontier orbitals.
Due to the poor stability of heavier group 14 elements with sp2-hybridization, the synthesis of the heavier analogues of simple compounds in carbon chemistry is not always simple. For instance benzene (C6H6) is an air- & moisture- stable aromatic compound. However, when the heavy atoms are incorporated into the ring system, the chemical property becomes totally different from that of C6H6. In this paper, Scheschkewitz group reports that the global minimum isomers of Ge2Si4 and Sn2Si4 compounds show non-planar, propellane-type structures. For the Ge2Si4 compound, they also could identify another isomer which has a dismutational aromatic nature. Scheschkewitz describes that these chemistry will be extended to the application such as staffane-type materials.[1]
-
References
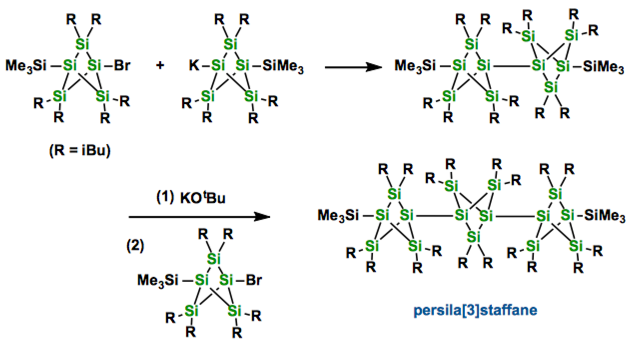
[1] “Persilastaffanes: Design, Synthesis, Structure, and Conjugation between Silicon Cages”
Iwamoto, T.; Tsushima, D.; Kwon, E.; Ishida, I.; Isobe, H. Angew. Chem. Int. Ed., 2012. 51, 2340 – 2344. DOI: 10.1002/anie.201106422
By stepwise catenation of bicyclo[1.1.1]pentasilane units persila[n]staffanes (n=1, 2, and 3) were synthesized as air-stable colorless crystals. A remarkable red-shift of the UV/Vis absorption bands with increasing number of bicyclo[1.1.1]pentasilane units suggests remarkable interactions between bridgehead SiSi σ-orbitals and between cage SiSi σ-orbitals
-
Related Links
Scheschkewitz Lab home Page
Iwamoto Lab home Page