Koichi Tanaka (born 1959, Toyama, Japan) is a Japanese analytical chemist and engineer, who shared the Nobel Prize in Chemistry in 2002 for developing a novel method for mass spectrometric analyses of biological macromolecules with John Bennett Fenn and Kurt Wüthrich (the latter for work in NMR spectroscopy).
He is the twelfth Japanese individual to receive the Nobel Prize and the first post-war- born Laureate. He was 43 years old when the award was announced, which makes him the second youngest after Prof. Hideki Yukawa, who was 42 when he was awarded the Nobel Prize in Physics in 1949. Moreover, Tanaka is the first ever person to win the Nobel Prize without a master’s or doctor’s degree, having become a researcher after graduating from university without entering a graduate school.
Currently, he is a core Researcher and general manager, Koichi Tanaka Laboratory of Advanced Science and Technology, Shimadzu Corporation.
-
Education and Experiences
1983 B. S. Tohoku University
1983 Central Research Laboratory, Shimadzu Corporation
2002 Fellow, Shimadzu Corporation
2003 General Manager of Mass Spectrometry Research Laboratory in Shimadzu Corporation
2010 General Manager, Koichi Tanaka Laboratory of Advanced Science and Technology, Shimadzu Corporation
-
Awards and Honors
1989 Encouragement Award from the Mass Spectrometry Society of Japan
2002 Nobel Prize for Chemistry
2002 Person of Cultural Merit, Japan
2003 Order of Culture, Japan
-
Research
Currently, two of the most popular life-science ionization techniques, MALDI (matrix-assisted laser-desorption ionization) and ESI (electrospray ionization). Tanaka developed the principal MALDI and Bennett Fenn who shared Nobel Prize Chemistry in 2002 with Tananaka invented the method of ESI.
Development Matrix-Assisted Laser Desorption Ionization Mass Spectrometry:MALDI-MS
Mass spectrometry (MS) is a method for measuring the weight of molecules and analyzing their contents. The ability to separate molecules based on different size and charge was first described in 1912 by J.J. Thompson (Nobel Prize laureate in 1906 for investigations of the conduction of electricity by gases). In mass spectrometry, it is necessary to use some method to separate the generated ions according to mass, and then perform detection of the ions.
“If you don’t ionize or you can’t ionize, then you can’t do mass spectrometry,”
Therefore, various ionization methods have been developed such as chemical ionization (CI), fast atom bombardment (FAB), and liquid matrix secondary ion mass spectrometry (LSIMS). Despite years of intense MS development, the goal of analysing large macromolecules remained elusive for over 70 years.
During the 1980s several groups tried to solve the volatilisation/ionisation problem of mass spectrometry using laser light as an energy source.
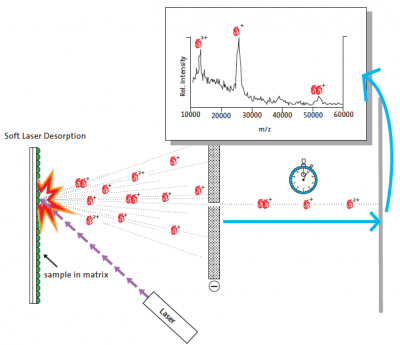
A breakthrough for the laser desorption method in its application to large biomolecules was reported at a symposium in Osaka in 1987, when Koichi Tanaka at the Shimadzu Corp. in Kyoto presented results of a mass spectrometric analysis of an intact protein.[1] The principle of soft laser desorption (SLD) is illustrated in Figure 2, showing the signals from singly and doubly charged molecular ions and a protein cluster-ion with a single charge.
The soft laser desorption process (adapted from NobelPrize.org)
A fast-growing version of the SLD technique, currently predominant, incorporates the macromolecules of interest in a low-molecular-weight crystalline matrix with absorption maximum matched to the wavelength of the laser pulse. This matrix-assisted, laser-desorption ionisation (MALDI) technique applied to proteins by Karas and Hillenkamp [16] that appeared shortly after Tanaka’s initial breakthrough.
In MALDI, there is a mixture of two different substances: the matrix and the analyte. The analyte is the substance of interest that needs to be ionized. The matrix is a substance that surrounds the analyte and assists in the ionization process. The matrix and the analyte are mixed together, usually in the liquid phase. The mixture is added, drop wise, to a metal plate. The solvent is allowed to evaporate, and the matrix crystallizes (forms a solid) around the analyte. The metal plate (which is usually stainless steel) is inserted in the instrument. A laser is shot at the sample, and ionization of the analyte is promoted. The ions travel through the instrument, and a mass spectrum is recorded. There are a variety of different types of matrix molecules; usually it is a small, organic acid. One of the most common matrices is called α-cyano-4-hydroxycinnamic acid.
The type of a mass spectrometer most widely used with MALDI is theTime-of-Flight Mass Spectrometry(TOF), which involves separating ions according to mass by measuring their respective flight times., mainly due to its large mass range.
LAMS-50K, the world’s first MALDI-TOF MS instrument (1988) form Shimazu Corp.
AXIMA: MALDI-TOF mass spectrometry from Shimazu Corp.
Currently, MS is widely used in various fields of science and technology, as it is an extremely effective method to measure the base quantity of compounds, both natural and artificial, and to identify them.
-
References
[1] Tanaka, K.; Waki, H.; Ido, Y.; Akita, S.; Yoshida, Y.; Yoshida, T. Rapid Commun. Mass Spectrom. 1988, 2, 151. DOI:10.1002/rcm.1290020802
[2] Karas, M. and Hillenkamp, F. Anal. Chem. 1998, 60, 2299. DOI: 10.1021/ac00171a028
-
Related Links
Koichi Tanaka - Wikipedia
Koichi Tanaka - Nobel Lecture: The Origin of Macromolecule Ionization by Laser Irradiation
[KOICHI TANAKA]a noble soul : SHIMADZU (Shimadzu Corporation)
-
Photo Gallery
-
Related Books
[amazonjs asin=”3527333312″ locale=”US” title=”MALDI MS: A Practical Guide to Instrumentation, Methods and Applications”][amazonjs asin=”0471775797″ locale=”US” title=”MALDI Mass Spectrometry for Synthetic Polymer Analysis”]