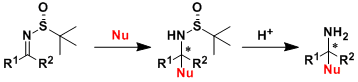Morales, S.; Guijarro, F. G.; García Ruano, J. L.; Cid, M. B. J. Am. Chem. Soc. 2014, ASAP.
DOI: 10.1021/ja4111418
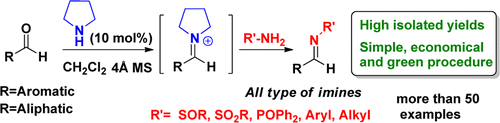
A general and efficient biomimetic method for the synthesis of aldimines from aldehydes and compounds bearing the NH2 group in the presence of pyrrolidine as a catalyst has been developed. These organocatalytic reactions, based on the application of the concept of nucleophilic catalysis, proceed with outstanding yields in the absence of acids and metals under simple conditions and minimum experimental manipulation. The method has been mainly applied to the synthesis of N-sulfinyl and N-sulfonyl imines, but its general validity has been proven with the preparation of representative N-phosphinoyl, N-alkyl, and N-aryl imines. These unprecedented reactions, which presumably occur via iminium activation without requiring acidic conditions, are an interesting and competitive alternative to the classical methods for preparing aldimines.
Well, I was very surprised to see this paper. Is this new method? In this paper, the authors, Cid and Ruano et al, described “To our knowledge, aminocatalytic methods for the direct preparation of imines from aldehydes and amines have never been reported, which is surprising taking into account the great advances achieved in organocatalytic processes.”

Enantiomerically pure N-sulfinyl imines, such as Ellman’s Imine[1] are one of the types of activated imines most used in the asymmetric synthesis of primary amines because of the dual role played by the sulfinyl group as an activating and stereodirecting group and its easy removal. The method most commonly used to prepare these compounds is based on the reaction of aldehydes with optically pure RSONH2 (R = p-Tol or t-Bu) in the presence of a large excess of Ti(OEt)4 (usually 5 equiv), which activates the aldehyde and acts as a dehydrating reagent. In contrast to the methods reported to date, which are usually based on the activation of the carbonyl with a large amount of acid, their surprisingly unprecedented procedure takes place under smooth catalytic conditions in the presence of pyrrolidine (10 mol %) and 4 Å MS, providing excellent yields of aldimines from a wide variety of aldehydes (aryl, heteroaryl, alkyl, and unsaturated) .
The asymmetric synthesis of amines as a chiral ammonia equivalent
- References
[1] “Synthesis and Applications of tert-Butanesulfinamide”
Robak, M. T.; Herbage, M. A.; Ellman, J. A. Chem. Rev. 2010, 110, 3600. DOI: 10.1021/cr900382t
- Related Links
Ruano group
- Related Books
[amazonjs asin=”3846580953″ locale=”US” title=”Picolylamine Template Organocatalysts for Asymmetric Aldol reactions: Pyridine based Organocatalysts for Asymmetric Aldol Reactions”][amazonjs asin=”1248945840″ locale=”US” title=”New nucleophilic organocatalysts.”]