Wang, P.; Dong, S.; Shieh, J.-H.; Peguero, E.; Hendrickson, R.; Moore, M. A. S.; Danishefsky, S. J. Science 2013, 342, 1357–1360.
DOI: 10.1126/science.1245095
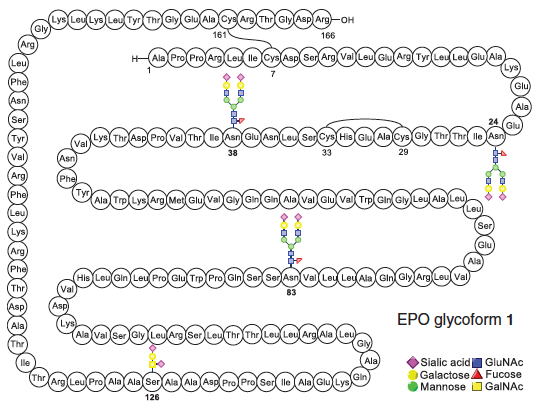
Erythropoietin is a signaling glycoprotein that controls the fundamental process of erythropoiesis, orchestrating the production and maintenance of red blood cells. As administrated clinically, erythropoietin has a polypeptide backbone with complex dishomogeneity in its carbohydrate domains. Here we describe the total synthesis of homogeneous erythropoietin with consensus carbohydrate domains incorporated at all of the native glycosylation sites. The oligosaccharide sectors were built by total synthesis and attached stereospecifically to peptidyl fragments of the wild-type primary sequence, themselves obtained by solid-phase peptide synthesis. The glycopeptidyl constructs were joined by chemical ligation, followed by metal-free dethiylation, and subsequently folded. This homogeneous erythropoietin glycosylated at the three wild-type aspartates with N-linked high-mannose sialic acid–containing oligosaccharides and O-linked glycophorin exhibits Procrit-level in vivo activity in mice.
EPO, is a glycoprotein hormone that controls erythropoiesis, or red blood cell production. It is a cytokine (protein signaling molecule) for erythrocyte (red blood cell) precursors in the bone marrow. Human EPO has a molecular weight of 34 kDa (consisting of a folded chain of 166 amino acids with carbohydrates attached at well-defined sites along the chain).Prof. Danishefsky and coworkers prepared an EPO sample by a chemical synthesis that maintains a uniform pattern of attached sugars throughout, which may prove helpful in the analysis of how variation in the sugar components of EPO impact function. Perspective of the chemistry: Improving Biologic Drugs via Total Chemical Synthesis
- Related Links
Samuel Danishefsky
- Related Books
[amazonjs asin=”0199257388″ locale=”US” title=”Amino Acid and Peptide Synthesis (Oxford Chemistry Primers, 7)”][amazonjs asin=”3642340644″ locale=”JP” title=”Total Synthesis of Natural Products: At the Frontiers of Organic Chemistry”]