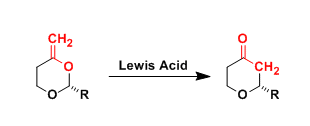
General Characteristics The Lewis acid-promoted rearrangement of enol acetals to cyclic ethers, which involves an oxygen-to-carbon transposition, is known as the Petasis-Ferrier rearrangement. This ...
Posts by Category: Reactions
Ferrier Rearrangement
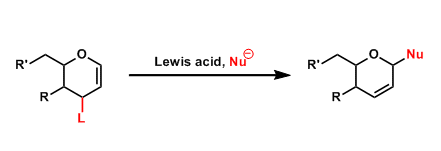
General Characteristics The introduction of nucleophilic groups to the 1-position of 1,2-glycals by an allylic substitution is called the Ferrier reaction. This reaction is referred to as the “Type ...
Ramberg-Backlund Rearrangement
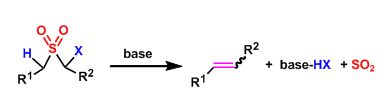
General Characteristics The basic treatment of α-halogenated sulfones leads to the formation of alkenes with the elimination of sulfur dioxide. This reaction is called the Ramberg-Backlund reaction ...
Benzidine Rearrangement
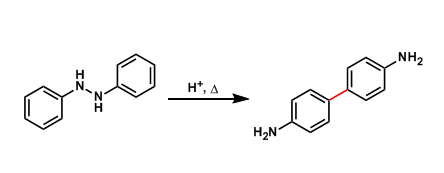
General Characteristics Under acidic conditions, 1,2-diphenylhydrazines are known to rearrange into 4,4’-diaminobiphenyls (benzidines). General References Hofmann, A. W. Proc. Roy. Soc. London ...
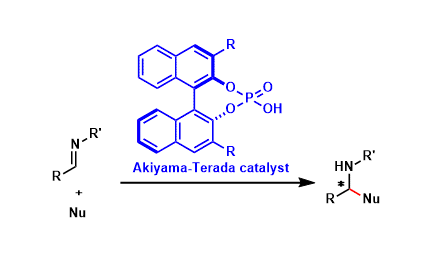
Akiyama-Terada Catalyst
General Characteristics The phosphoric acids containing an appropriately substituted BINOL backbone function as chiral Brønsted acid catalysts that can promote a wide range of stereoselective ...
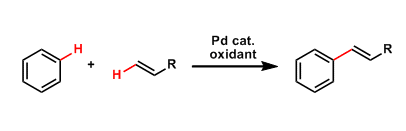
Fujiwara-Moritani Reaction
General Characteristics The direct coupling between unsubstituted aromatic rings and olefins in the presence of palladium catalysts is called the Fujiwara-Moritani reaction. This reaction is one of ...
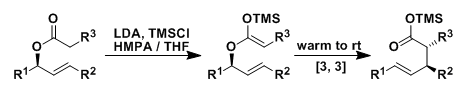
Ireland-Claisen Rearrangement
General Characteristics The Ireland-Claisen rearrangement is a version of the Claisen rearrangement in which ketene silyl acetals (prepared from allyl esters) undergo [3,3]-sigmatropic rearrangement ...
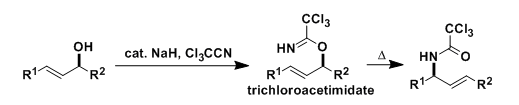
Overman Rearrangement
General Characteristics The trichloroacetimidates of allylic alcohols, prepared using trichloroacetonitrile, undergo thermal or catalyzed [3,3]-sigmatropic rearrangement, which is referred to as the ...
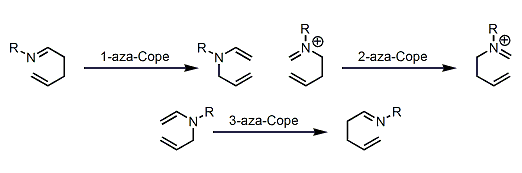
Aza-Cope Rearrangement
General Characteristics Nitrogen-containing 1,5-dienes undergo [3,3]-sigmatropic rearrangement just like the Cope rearrangement of 1,5-dienes. Among these so-called aza-Cope rearrangements, the 3-aza ...
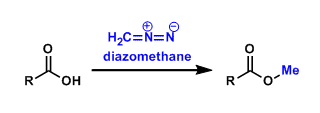
Diazomethane
General Characteristics Diazomethane (CH2N2) is the simplest example of diazo compounds. It is traditionally used to synthesize methyl esters of carboxylic acids. This methyl ester formation is very ...