Wang, Q.; Motika, S. E.; Akhmedov, N. G.; Petersen, J. L.; Shi, X. Angew. Chem. Int. Ed. 53, 2014. 53, 5418.
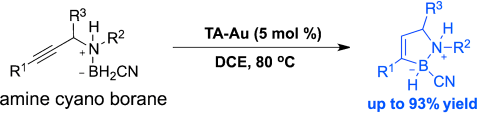
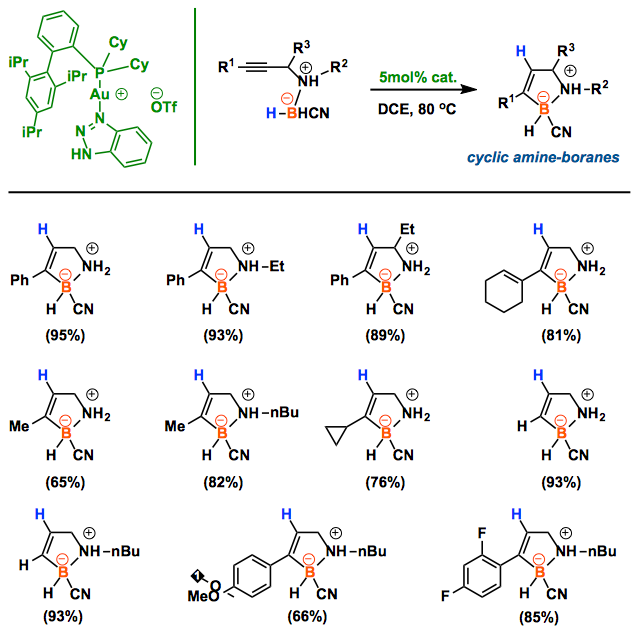
The first catalytic alkyne hydroboration of propargyl amine borane carbonitriles is accomplished with triazole-Au complexes. While the typical [L-Au]+species decomposes within minutes upon addition of amine borane carbonitriles, the triazole-modified gold catalysts (TA-Au) remained active, and allowed the synthesis of 1,2-BN-cyclopentenes in one step with good to excellent yields. With good substrate tolerability and mild reaction conditions (open-flask), this new method provides an alternative route to reach.
During the past decade, cationic gold complexes have been recognized as one of the most useful catalysts to activate CC multiple bonds due to their soft p-acidity. To the best of my knowledge, however, there is no report about the gold catalysis using amine-borane as the substrate. This is because the cationic gold is readily reduced by amine-borane which results in the decomposition of the catalyst with the formation of the Au(0) particles. Probably, that’s why gold complexes have never been used as the catalysts for the dehydrogenation reaction of ammonia-borane, despite the rapid progress of the research with the other transition metals based on the high feasibility of ammonia-borane as a H2 storage material.
Recently Xiaodong group has demonstrated that the stability of gold complex can be extremely increased by the incorporation of 1,2,3-triazole (TA) into cationic gold center.[1] In this paper, they reported that a catalytic amount of a gold-TA complex system promoted the intramolecular hydroboration of alkyne with amine-borane, which allows for the one-pot synthesis of the cyclic amine-borane derivatives. Significantly, the overall transformation operates under open-flask conditions. This Au-TA system will be applied to prepare other BN/CC isosteric derivatives such as benzene and indole too.
-
References
[1] “1,2,3-Triazole as a Special “X-Factor” in Promoting Hashmi Phenol Synthesis”
Chen, Y.; Wuming Yan, W.; Akhmedov, N. G.; Shi, X. Org. Lett. 2010, 12, 344 – 347. DOI: 10.1021/ol902680k
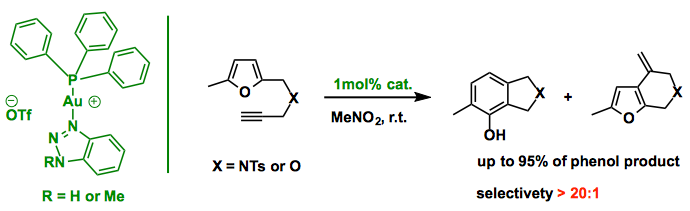
31P NMR experiments revealed rapid decomposition of the Ph3PAu+TfO–complex in the presence of the furan-yne, which resulted in poor reactivity as reported in the literature. Therefore, instead of tuning different ligands (PR3), the 1,2,3-triazole was applied as a special X-factor to stabilize the catalyst. The desired phenol products were prepared in excellent yields (1% cat. up to 95% yield) and chemoselectivity (>20:1).
-
Related Links