Fuse, S.; Mifune, Y.; Takahashi, T. Angew. Chem. Int. Ed. 2013, Early View
DOI: 10.1002/anie.201307987
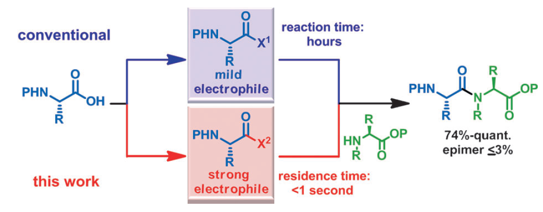
The development of highly efficient amide bond forming methods which are devoid of side reactions, including epimerization, is important, and such a method is described herein and is based on the concept of rapid and strong activation of carboxylic acids. Various carboxylic acids are rapidly (0.5 s) converted into highly active species, derived from the inexpensive and less-toxic solid triphosgene, and then rapidly (4.3 s) reacted with various amines to afford the desired peptides in high yields (74 %–quant.) without significant epimerization (≤3 %). Our process can be carried out at ambient temperature, and only CO2 and HCl salts of diisopropylethyl amine are generated. In the long history of peptide synthesis, a significant number of active coupling reagents have been abandoned because the highly active electrophilic species generated are usually susceptible to side reactions such as epimerization. The concept presented herein should renew interest in the use of these reagents.
Microreactors have received significant interest in the stream of downsizing of chemistry, and they are expected to make an innovative and revolutionary change for chemical synthesis.In addition to the safety and environmental benefits, micro flow reactors have several advantages over conventional systems stemming from the high surface-to-volume ratio; e.g. precise temperature control and high efficiency of heterogeneous mass transfer. Extremely fast mixing by virtue of short diffusion path is also noteworthy. A short residence time in reactors may also be advantageous from a viewpoint of the control of highly reactive intermediates.
Dr. Shinichiro Fuse, assistant professor at Tokyo Institute of Technology in Japan, demonstrated highly efficient amide bond formation based on our concept of rapid and strong activation of carboxylic acids by using a micro flow reactor.
- Related Books
[amazonjs asin=”3319029185″ locale=”US” title=”Controlled Polymerization and Polymeric Structures: Flow Microreactor Polymerization, Micelles Kinetics, Polypeptide Ordering, Light Emitting Nanostructures (Advances in Polymer Science)”][amazonjs asin=”3527332820″ locale=”JP” title=”Microreactors in Preparative Chemistry: Practical Aspects in Bioprocessing, Nanotechnology, Catalysis and more”]