X, Xiong.; F, Song.; J, Wang.; Y, Zhang.; Y, Xue.; L, Sun.; Na Jiang.; P, Gao.; L, Tian.; X, Peng, J. Am. Chem. Soc. 2014, 136, 9590–9597.
DOI: 10.1021/ja502292p
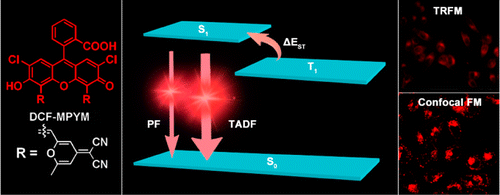
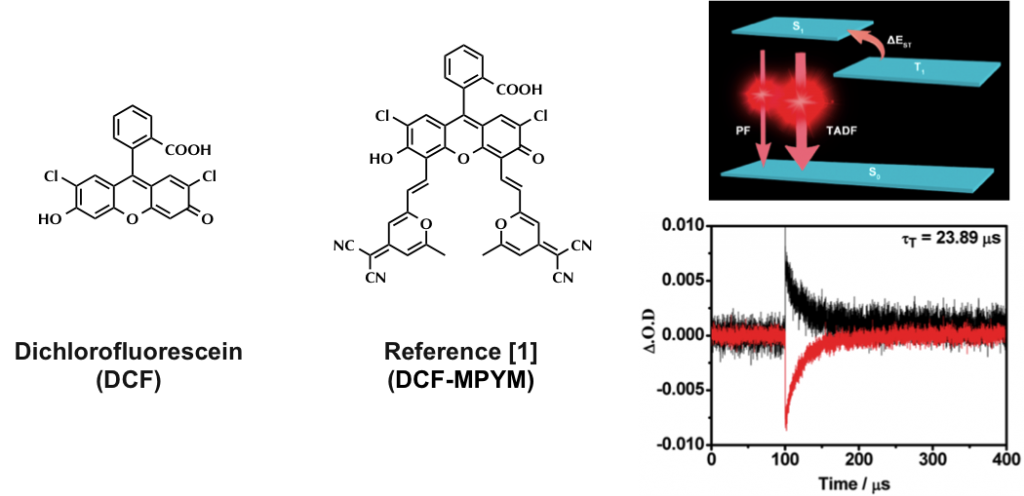
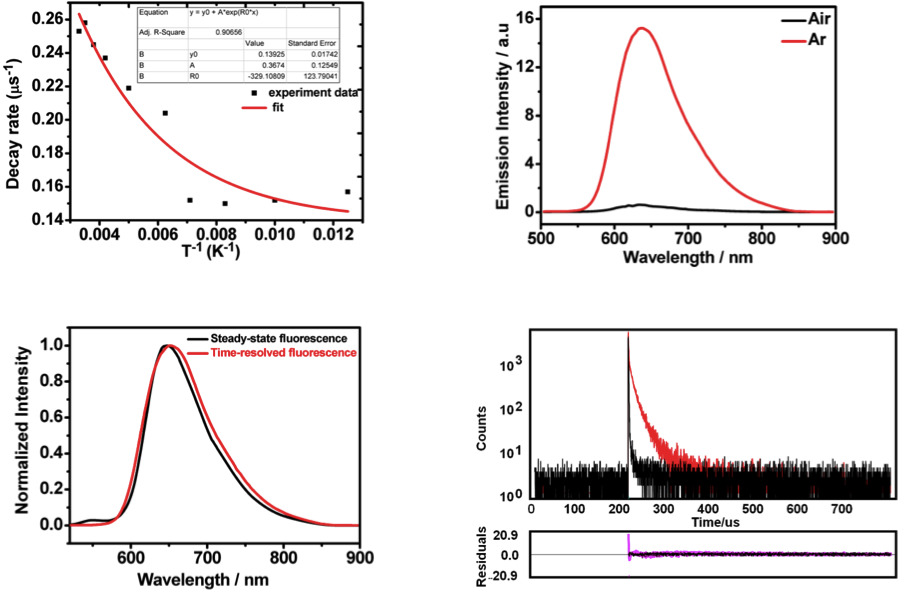
Compared with fluorescence imaging utilizing fluorophores whose lifetimes are in the order of nanoseconds, time-resolved fluorescence microscopy has more advantages in monitoring target fluorescence. In this work, compound DCF- MPYM, which is based on a fluorescein derivative, showed long-lived luminescence (22.11 μs in deaerated ethanol) and was used in time-resolved fluorescence imaging in living cells. Both nanosecond time-resolved transient difference absorption spectra and time-correlated single-photon counting (TCSPC) were employed to explain the long lifetime of the compound, which is rare in pure organic fluorophores without rare earth metals and heavy atoms. A mechanism of thermally activated delayed fluorescence (TADF) that considers the long wavelength fluorescence, large Stokes shift, and long-lived triplet state of DCF-MPYM was proposed. The energy gap (ΔEST) of DCF-MPYM between the singlet and triplet state was determined to be 28.36 meV by the decay rate of DF as a function of temperature. The ΔEST was small enough to allow efficient intersystem crossing (ISC) and reverse ISC, leading to efficient TADF at room temperature. The straightforward synthesis of DCF-MPYM and wide availability of its starting materials contribute to the excellent potential of the compound to replace luminescent lanthanide complexes in future time-resolved imaging technologies.
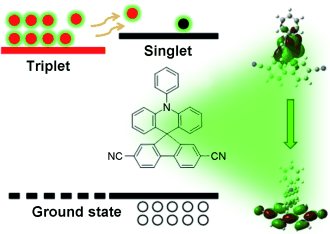
Radiative deactivation with long lifetime (~μs) is very powerful tool of visualizing biological activities. To do so, phosphorescence from triplet state was extensively studied so far. Despite much effort, however, such sensitizers often encounter unavoidable defects, such as generation of reactive oxygen and high cytotoxicity. The authors of the paper put through fluorescein derivatives exhibiting long lifetime fluorescence, activated delayed fluorescence (TADF).
TADF: activated delayed fluorescence
To confirm the long lifetime emission is attribute to TADF, the lifetime over temperature change and the effect of oxygen was measured.[2] As shown below (left top figure), fluorescence lifetime was evidently elongated when the temperature rises. In addition, the spectrum of time-resolved fluorescence shape and that of steady state are almost identical (left bottom figure). Also, fluorescence was effectively bleached by the presence of oxygen and the lifetime of which is also lowered (right top and bottom figure). Although not shown in detail, quantum calculation also revealed that the electron distribution of HOMO and LUMO were well separated. All these experimental results suggest that the long lifetime emission is due to TADF.
Energy diagram
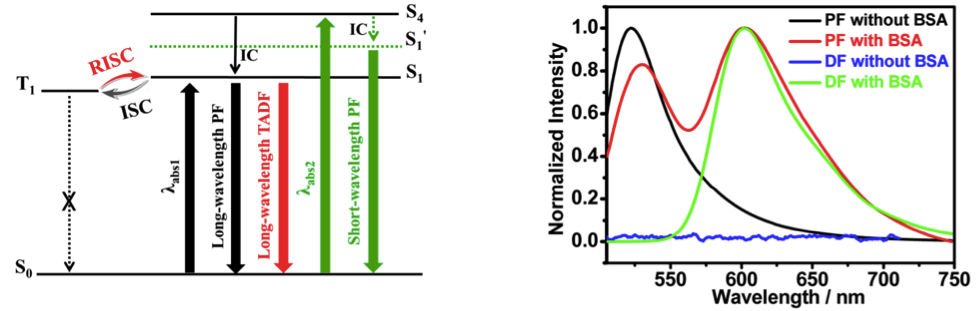
Based on above experimentation, authors hypothesized energy diagram that explains the emission behavior of this dye. In protic environment, electrons are predominantly excited to higher excited states than S1, which accordingly results in shorter and higher energy fluorescence. When surrounded by apotic atmosphere, electrons mainly populate the S1 state. The presence of T1 state that has almost same energy level relative to S1 helps repopulate the S1 state that leads to following the delayed fluorescence.
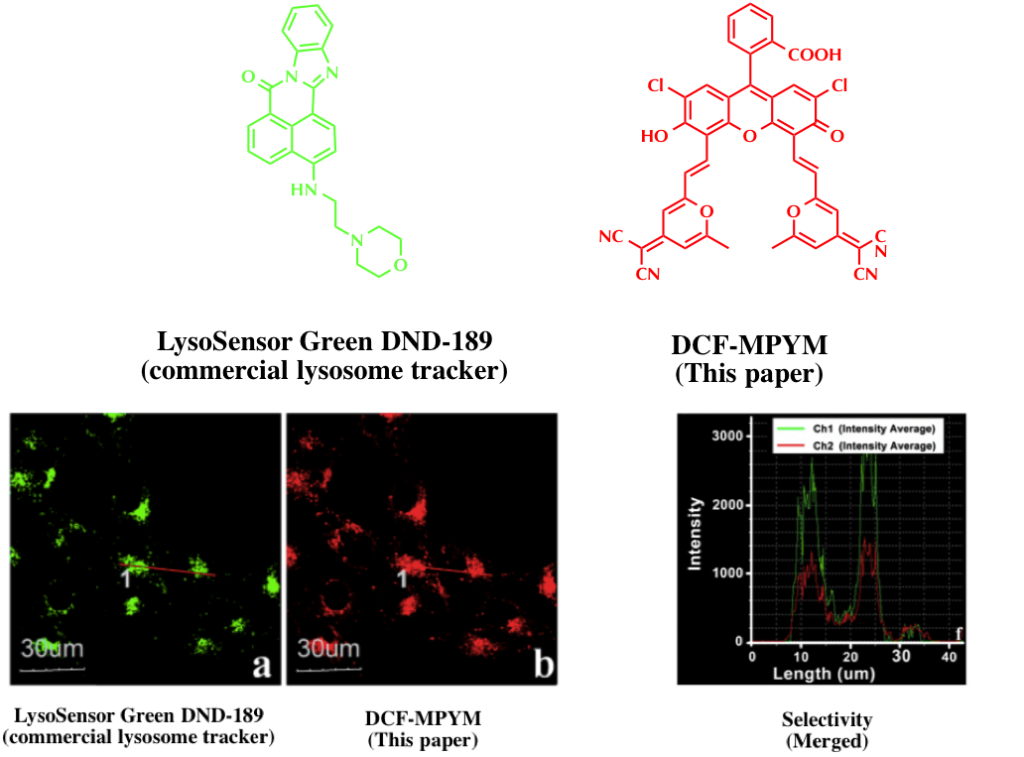
Cell imaging
DCM-MPYM localized in lysosome with high selectivity and its TADF was used for detection. The selectivity was confirmed by multicolor imaging experiment using LysoSensor Green DND-189, a commercial dye for lysosome, together with DCM-MPYM. By exploiting same strategy in this paper, dye with various TADF colors and with various organelle selectivity may be realized.
References
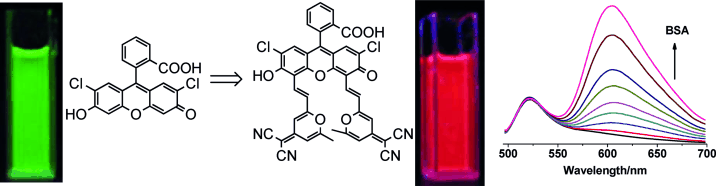
[1] “Red-Emissive Fluorescein Derivatives and Detection of Bovine Serum Albumin”
Xiaoqing Xiong , Fengling Song *, Jingyun Wang , Yukang Zhang , Yingying Xue , Liangliang Sun , Na Jiang , Pan Gao , Lu Tian , and Xiaojun Peng. Asian. J. Or.g Chem. 2013, 2, 145. DOI: 10.1002/ajoc.201200109
Red light: Two fluorogenic compounds were synthesized by derivatization of 2′,7′-dichlorofluorescein at the C4′ and C5′ positions. These compounds have excellent photostability and emit at red wavelengths. Both compounds are highly selective for detecting bovine serum albumin because of their particular dyad structures.
[2] “Enhanced Electroluminescence Efficiency in a Spiro-Acridine Derivative through Thermally Activated Delayed Fluorescence”
Mehes, G.; Nomura, H.; Zhang, Q.; Nakagawa, T.; Adachi, C. Angew. Chem., Int. Ed. 2012, 51, 11311. DOI: 10.1002/anie.201206289
Make your OLED fluorescent: An aromatic molecule based on a spiro-acridine derivative was designed (see picture), and its photoluminescence and electroluminescence were characterized. By combining the donor and acceptor moieties a small energy gap between the lowest singlet and triplet states was achieved. This design leads to an organic light-emitting diode (OLED) that rivals phosphorescent devices regarding exciton generation efficiency.
Related Books
[amazonjs asin=”0857094254″ locale=”US” title=”Organic Light-Emitting Diodes (OLEDs): Materials, Devices and Applications (Woodhead Publishing Series in Electronic and Optical Materials)”]