- Generality
- Reagent Availability
- Experimental User Friendliness
- Criteria #4
- Criteria #5
-
General Characteristics
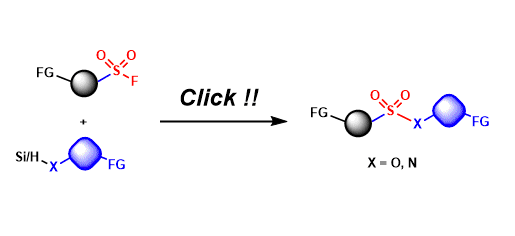
Sulfur(VI) fluoride compounds (e.g. RSO2F, SO2F2) are stable under a wide range of conditions, yet their S-F bonds can be activated for exchange reactions in the presence of proton or silicon reagents.
Thanks to the high chemoselectivity and functional group tolerance, sulfur-fluoride exchange (“SuFEx”) is proposed as a new reaction for click chemistry.
-
General References
<Reviews>
- Steinkopf, W. J. Prakt. Chem. 1927, 1, 1. DOI: 10.1002/prac.19271170101
- Dong, J.; Krasnova, L.; Finn, M. G.; Sharpless, K. B. Angew. Chem. Int. Ed.2014, Early View. DOI: 10.1002/anie.201309399
<Click chemistry>
- Kolb, H. C.; Finn, M. G.; Sharpless, K. B. Angew. Chem. Int. Ed. 2001, 40, 2004. [abstract]
-
Reaction Mechanism
Sulfur(VI) fluorides generally exist as stable compounds for the following reasons.
The bond energy of S(VI)-F is about 80-90 kcal/mol (SO2F2), which is much higher than that of S(VI)-Cl (SO2Cl2: 46 kcal/mol).
The nucleophilic addition-elimination reactions at S(VI) centers are relatively slow. For example, the Nu-Cl byproduct is sometimes observed in reactions between RSO2Cl and a nucleophile, which is considered to occur as a result of the nucleophile attacking the chloride instead of the sulfur center.
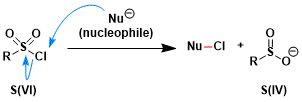
The activation of stable sulfur(VI) fluorides for exchange reactions requires assistance from proton (H+) or silicon (R3Si+).
-
Examples
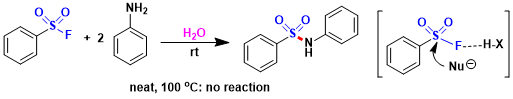
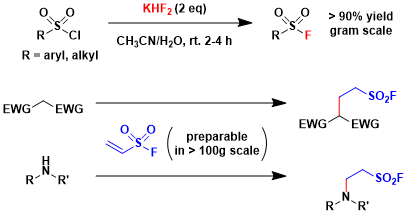
Preparation of sulfonyl fluorides: Sulfonyl chlorides can be easily converted to the corresponding fluorides. Alkyl sulfonyl fluorides can be synthesized by the Michael addition of various nucleophiles to vinyl sulfonyl fluoride.
Gaseous SO2F2 and ROH react to form fluorosulfates. Since –OSO2F is a good leaving group, the application to functionalize phenols is common. Sulfamoyl fluorides are synthesized similarly from SO2F2 and secondary amines. The products formed from primary amines tend to be unstable due to the elimination of HF.
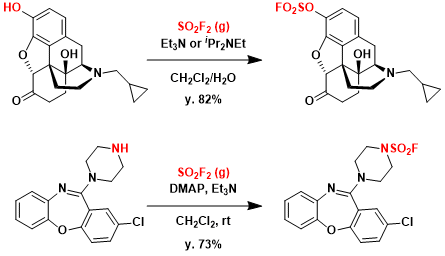
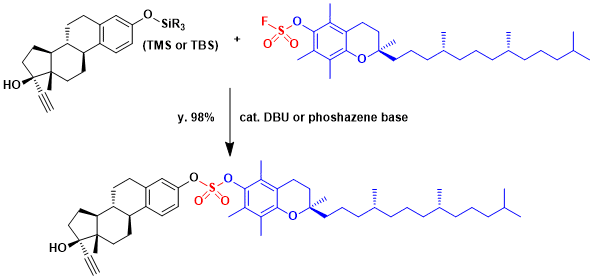
The exchange reaction with silyl protected phenols in the presence of a catalytic amount of base is also known. As shown below, it can couple two very large fragments. The silyl fluoride byproduct is unreactive and thus minimally interferes with the reaction.
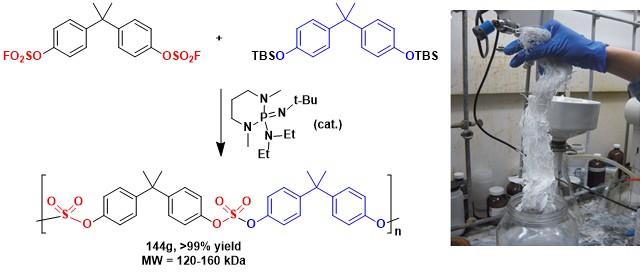
The synthesis of bisphenol A sulfate polymer by SuFEx.[1]
-
Experimental Procedure
-
Experimental Tips
The toxic and gaseous SO2F2 should be handled carefully under well-ventilated fume hoods.
-
References
[1] Dong, J.; Sharpless, K. B.; Kwisnek, l.; Oakdale, J. S.; Fokin, V. V. Angew. Chem. Int. Ed. 2014, Early View. doi:10.1002/anie.201403758
-
Related Reactions
-
Related Books
[amazonjs asin=”0470699701″ locale=”US” title=”Click Chemistry for Biotechnology and Materials Science”]
[amazonjs asin=”3527320857″ locale=”US” title=”Click Chemistry: In Chemistry, Biology and Macromolecular Science”]
-
External Links