Chenguang Wang, Masayasu Taki, Yoshikatsu Sato, Aiko Fukazawa, Tetsuya Higashiyama, and Shigehiro Yamaguchi. J. Am. Chem. Soc., 2017, 139, 10374–10381.
DOI: http://10.1021/jacs.7b04418
Abstract
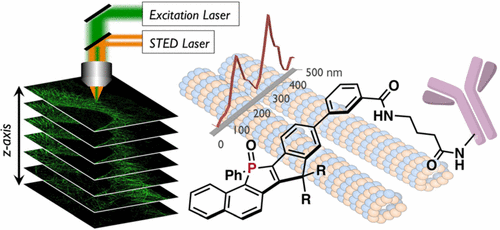
As stimulated emission depletion (STED) microscopy can provide structural details of cells with an optical resolution beyond the diffraction limit, it has become an indispensable tool in cell biology. However, the intense STED laser beam usually causes rapid photobleaching of the employed fluorescent dyes, which significantly limits the utility of STED microscopy from a practical perspective. Herein we report a new design of super-photostable dye, PhoxBright 430 (PB430), comprising a fully ring-fused π-conjugated skeleton with an electron-accepting phosphole P-oxide unit. We previously developed a super-photostable dye C-Naphox by combining the phosphole unit with an electron-donating triphenylamine moiety. In PB430, removal of the amino group alters the transition type from intramolecular charge transfer character to π–π* transition character, which gives rise to intense fluorescence insensitive to molecular environment in terms of fluorescence colors and intensity, and bright fluorescence even in aqueous media. PB430 also furnishes high solubility in water, and is capable of labeling proteins with maintaining high fluorescence quantum yields. This dye exhibits outstanding resistance to photoirradiation even under the STED conditions and allows continuous acquisition of STED images. Indeed, using a PB430-conjugated antibody, we succeed in attaining a 3-D reconstruction of super-resolution STED images as well as photostability-based multicolor STED imaging of fluorescently labeled cytoskeletal structures.
Introduction
Fluorescence nanoscopy, especially STED microscopy, is increasingly used for the visualization of biological samples, and also for the examination of various physical and chemical processes.[1] However, the necessity to irradiate samples with a donut-shaped depletion laser beam (STED laser) with extremely high power (>10 MW/cm2) severely limits STED microscopy from a practical perspective. Despite numerous efforts have been made to increase the photo-resistance of dyes, the advent of highly photo-resistant fluorescent dye for STED is still highly demanded. In this paper, Prof. S. Yamaguchi at Nagoya Univ. reported the design of the second-generation super-photostable fluorescent dye PhoxBright 430,[2] denoted as PB430. PB430 significantly expands the practical utility of such fluorescent dyes in STED nanoscopy.
Details
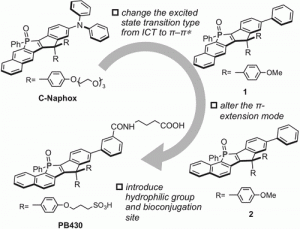
Figure 1 shows the molecular design of PB430 based on the rational structural modification of the first-generation super-photostable dye C-Naphox.
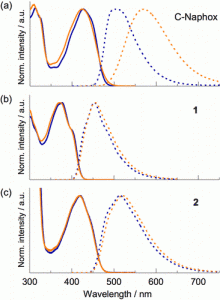
Figure 2 shows the normalized absorption (solid line) and fluorescence (dashed line) spectra of (a) C-Naphox, (b) 1, and (c) 2 in toluene (blue line) and CH3CN (orange line).
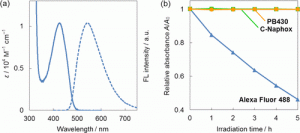
Figure 3 shows the (a) absorption (solid line) and fluorescence (dashed line) spectra of PB430 in PBS buffer (pH 7.4) and (b) comparison of the photostability among Alexa Fluor 488, C-Naphox, and PB430. Absorption variation of DMSO/HEPES buffer solutions (pH = 7.3, v/v = 7/3) of Alexa Fluor 488 (blue triangles), C-Naphox (green squares), and PB430 (orange circles) were plotted as a function of irradiation time with a Xe lamp (9 mW cm–2) equipped with a band-pass filter of 460 ± 11 nm. Solution concentrations were adjusted to be comparable to one another in terms of optical density at 460 nm.
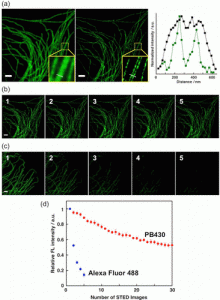
Figure 4 shows fluorescence images of immunolabeled-microtubules in a fixed HeLa cell: (a) Confocal (left) and STED (middle) microscopy images and the intensity profiles (right) along with the dotted lines in the insets of the confocal (black line) and STED (green line) images. (b, c) First five STED microscopy images stained with (b) PB430 and (c) Alexa Fluor 488. (d) Integrated fluorescence intensity plotted as a function of the number of recorded STED images. All images were recorded with the excitation at 470 nm and a depletion laser of 592 nm (CW-STED, 30 mW) was used for STED. Scale bars indicate 2 μm.
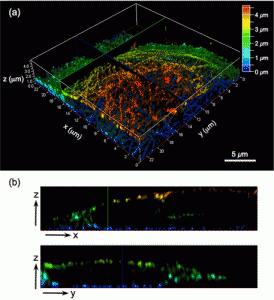
Figure 5 shows (a) 3-D STED image of HeLa cell microtubules immunolabeled with PB430, including a color scale corresponding to the height (increments in z-direction, 50 nm; scale bar, 5 μm). The images were recorded under the following STED conditions: excitation at 470 nm (WLL, 5 μW), depletion at 592 nm (CW-STED, 30 mW; STED 3D z donut, 50%), and gated detection at tg = 0.5 ns. Each image was deconvoluted using the Huygens Deconvolution Software (signal-to-noise ratio, 7; quality threshold, 0.05) and the z-sectioning images were colored to represent the z-depth. (b) STED images along the corresponding xz and yzplanes in panel a.
Reference
[1]”STED Microscopy Reveals that Synaptotagmin Remains Clustered after Synaptic Vesicle Exocytosis.”
Willig, K. I.; Rizzoli, S. O.; Westphal, V.; Jahn, R.; Hell, S. W. Nature 2006, 440, 935−939.
DOI: 10.1038/nature04592
[2]”A Phosphole Oxide Based Fluorescent Dye with Exceptional Resistance to Photobleaching: A Practical Tool for Continuous Imaging in STED Microscopy”
Wang, C.; Fukazawa, A.; Taki, M.; Sato, Y.; Higashiyama, T.; Yamaguchi, S. Angew. Chem., Int. Ed. 2015, 54, 15213−15217.