Reliable papers summarizing NMR data of various solvents in various heavy solvents are indispensable to organic chemists. In this post, four papers that contain NMR chemical shifts are presented:
Why Impuirites in NMR?
After purification in organic synthesis, the solvent is removed under reduced pressure, and NMR analysis is performed to confirm purity and determine the structure of the product. Sometimes, solvent remains, or impurities (lipids, plasticizers, silicone grease, etc.) are mixed in. In such cases, “papers summarizing NMR data of various solvents in various deuterated solvents” are very useful for identifying these residual solvents and impurities.
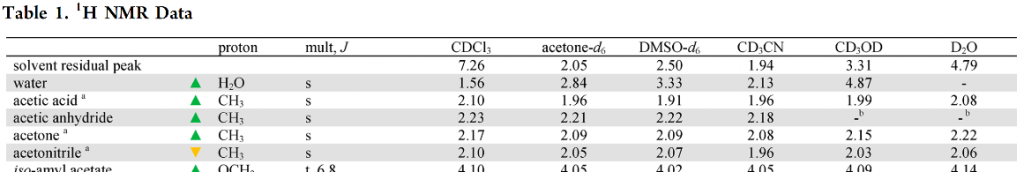
JOC 1997 ver[1]
The original NMR solvent paper. The author is Prof. Abraham Nudelman from Bar-Ilan University in Israel (medicinal chemistry, including drug delivery). Prof. Nudelman has been a professor at Bar-Ilan University since 1994 and a distinguished professor emeritus since 2008.
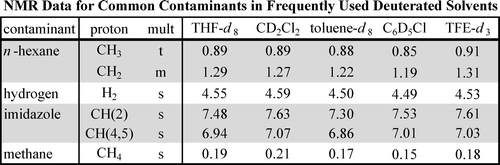
Organometallics 2010 ver[2]
The organometallic version published in 2010. New heavy solvents were added, including tetrahydrofuran-d8, toluene-d8, dichloromethane-d2, chlorobenzene-d5, and 2,2,2-trifluoroethanol-d3, which are used in the synthesis of organometallic complexes. The authors are from the University of Washington and the California Institute of Technology, as well as Bar-Ilan University in Israel, which authored the original JOC. The corresponding author is Prof. Karen I. Goldberg of the University of Washington, who specializes in organometallic complexes and has been a professor there since 2007.
Organic Process Research & Development 2016 ver[3]
A paper on modern NMR solvents published in 2016. Chemical shifts of environmentally benign solvents used in real industry, such as 2-Me-THF, n-heptane, and iso-propyl acetate, are included. The author is a team from Dow AgroSciences.
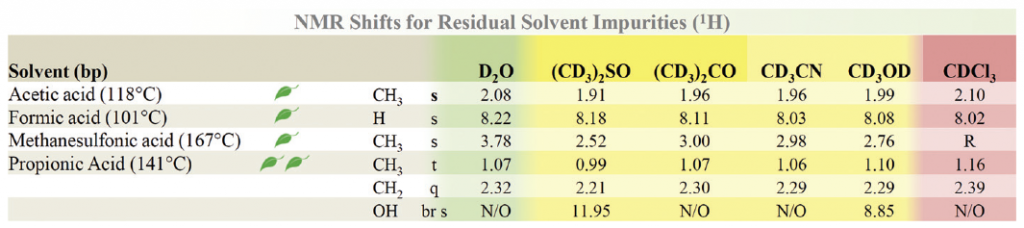
Green Chem 2016 ver[4]
The concept is a bit similar to that of the OPR&D paper. Data on solvents relevant to green chemistry are highlihgted. The authors are a team from GSK, a British pharmaceutical company, and C2M AUROCHS Industrie, a French synthesis company, plus Bar-Ilan University in Israel. It ranks the environmental impact of solvents based on the GSK Solvent Sustainability Guide.
Comparison
| No. of Deuterated solvents | No. of Impurities | |
| J. Org. Chem., 1997, 62, 7512–7515 | 7 | 35 |
| Organometallics, 2010, 29, 2176–2179 | 12 | 42 |
| Org. Process Res. Dev., 2016, 20, 661–667 | 6 | 51 |
| Green Chem., 2016,18, 3867-3878 | 6 | 80 |
References
- Gregory R.; Alexander Miller., Nathaniel H., Hugo E., Abraham N., Brian M., John E., Karen I. Organometallics, 2010, 29, 2176–2179. DOI:10.1021/jo971176v
- Nicholas R., Elizabeth O., Gregory T., Belgin C., Nakyen C., Lawrence C., Carl V., Nicole M., Peter L., James A., Fangzheng L., Beth A., Benjamin M., Sarah J., Michelle R., and Qiang Y. Org. Process Res. Dev., 2016, 20, 661–667. DOI:10.1021/acs.oprd.5b00417